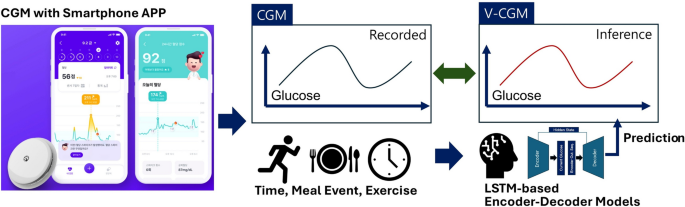
This study proposes a 'Virtual CGM' deep learning model that predicts current and future blood glucose levels using only life-log data such as food intake and exercise records, without actual blood glucose measurements. Developed from data of 171 healthy adults, this model combines an LSTM-based encoder-decoder architecture with food name embeddings and autocorrelation techniques to significantly improve prediction accuracy. It demonstrates the potential to serve as an intermittent monitoring alternative for both diabetes patients and healthy individuals seeking health management, addressing the high cost and discomfort of existing CGM devices.
1. Introduction: Why Do We Need 'Virtual' Glucose Monitoring?
Continuous Glucose Monitors (CGMs) enable real-time tracking of blood glucose changes and have gained attention not only for diabetes management but also for healthy individuals' dietary and weight management. However, significant barriers exist: expensive costs and the discomfort of wearing a needle under the skin.
The maintenance cost of CGM devices is relatively high, which can impose an economic burden on users during long-term use. [...] Therefore, technology that mimics actual CGM operation by utilizing several features of life-logs to infer current blood glucose levels has become necessary.
The research team developed a Virtual CGM model that predicts blood glucose using only life-log data (meals, exercise, etc.) recorded on smartphones or wearable devices. While existing blood glucose prediction models typically require 'recent blood glucose records,' this model's most distinctive feature is that it infers current and future blood glucose without any past blood glucose data, using only life-logs.
2. Methods: Data Collection and Model Design
The team collected data from 171 healthy adults between October 2023 and August 2024. Participants wore Dexcom G7 CGMs and logged meal (calories, nutritional content, food names) and exercise (step count, exercise intensity) information through a dedicated app.
2.1. Data Processing
Data was processed using the sliding window technique.
- Input: 90 minutes of past life-log data from the current time point (0 min) — meals, exercise, time information, etc. No past blood glucose data included.
- Output: Blood glucose changes from the current time point (0 min) to 90 minutes into the future.
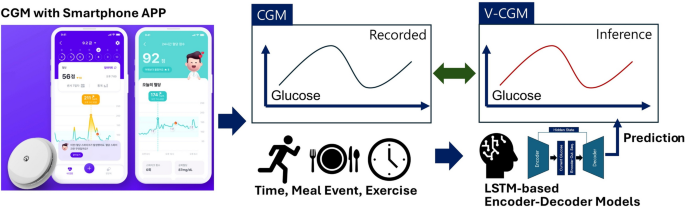 Figure 1: Overall study overview. Life-log data is collected, preprocessed, then blood glucose is inferred through the deep learning model (V-CGM).
Figure 1: Overall study overview. Life-log data is collected, preprocessed, then blood glucose is inferred through the deep learning model (V-CGM).
2.2. Deep Learning Model Architecture
The model adopts an encoder-decoder architecture based on LSTM (Long Short-Term Memory), which excels at processing time-series data. Several special techniques were added to boost performance.
- Attention Mechanism: Helps the model focus on which input elements (e.g., carbohydrate intake) have the greatest impact on blood glucose changes.
- Autocorrelation Module: Helps better capture periodic patterns in the data.
- Food Name Embedding (RoBERTa): Beyond simple 'grams of carbohydrates,' uses a natural language processing model to extract meaning from food names (text) themselves. For example, it can understand the difference between an 'apple' and a 'cake' even when the carbohydrate content is similar.
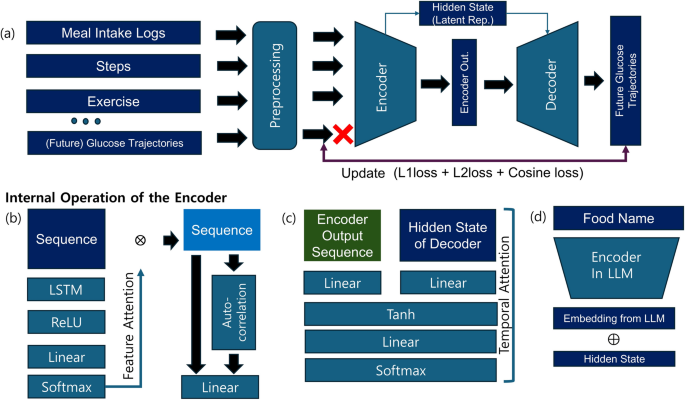 Figure 2: Model architecture diagram. The encoder analyzes life-logs and the decoder predicts blood glucose based on this analysis. Food name embedding and attention modules are combined.
Figure 2: Model architecture diagram. The encoder analyzes life-logs and the decoder predicts blood glucose based on this analysis. Food name embedding and attention modules are combined.
3. Results: How Accurate Were the Predictions?
The proposed model showed significantly meaningful prediction performance even without past blood glucose information.
3.1. Prediction Performance and Key Factors
- Current blood glucose (0 min) prediction: Mean Absolute Error (MAE) was approximately 14.93 mg/dL, achieving fairly close estimates without actual CGM.
- Power of personalized models: The best performance was achieved by first creating a 'universal model' trained on everyone's data, then fine-tuning it with each individual's data.
- Importance of food information: The most important data for model predictions was carbohydrates. Exercise volume and meal timing also played complex roles.
3.2. Attention Analysis Visualization
Visualizing where the model focused confirmed that it placed high weights on the timing of food intake and its nutritional components.
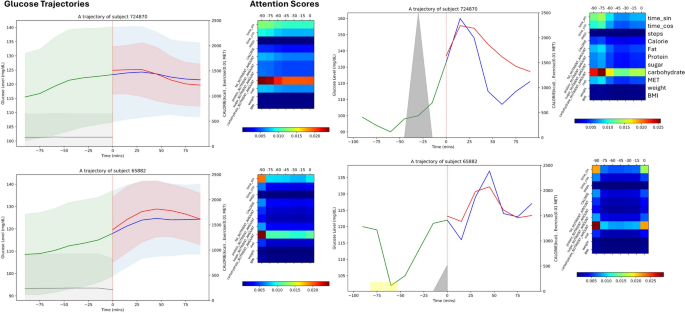 Figure 4: Comparison of predicted blood glucose (red) and actual blood glucose (blue). The model responds sensitively to meal events (gray) and predicts blood glucose rises.
Figure 4: Comparison of predicted blood glucose (red) and actual blood glucose (blue). The model responds sensitively to meal events (gray) and predicts blood glucose rises.
4. Discussion: Impact of This Technology on Our Lives
The use of food names (text) proved to be a masterstroke. Even when nutritional information was inaccurate or missing, approximate blood glucose responses could be inferred from food names alone. The autocorrelation module also played a major role in capturing short-term blood glucose variability.
However, the team expressed a cautious stance about using virtual CGM for healthy people.
If CGM use spreads to the general public without education and guidance, it may cause confusion rather than clarity. While CGM has promising applications beyond diabetes, its use by healthy individuals should be carefully investigated, supported by clear clinical frameworks and additional research.
In other words, while this technology offers major advantages in cost reduction and improved accessibility, it requires the prerequisite of accurate data entry by users and consideration of excessive health anxiety among healthy people.
5. Conclusion
This 2025 study demonstrated that while deep learning-based virtual CGM cannot fully replace actual devices, it can serve as an excellent supplementary tool for intermittent monitoring or addressing cost issues.
Key Takeaways:
- No Blood Required: Blood glucose prediction possible from life-logs alone, without past blood glucose data.
- Smart AI: Improved accuracy by combining LSTM, attention, and natural language processing (food names).
- Cost-Effective: Indirect blood glucose management possible without continuously wearing expensive CGM sensors.
If this technology is combined with diabetes patient data and more diverse biosignals like sleep and stress, we may usher in the era of needle-free blood glucose management sooner than expected.