This paper presents a study that developed and validated an algorithm for non-invasively measuring blood glucose using wearable photoplethysmography (PPG) sensors and machine learning, and for assessing diabetes risk based on repeated measurements. The key contribution is overcoming the limitations of existing invasive methods and confirming the potential for broader and easier diabetes risk screening and monitoring. The core conclusion is that an SVM-based algorithm effectively detects elevated blood glucose with an average accuracy of 84.7%, and this approach can support population-level early diabetes screening.
1. Research Background and Problems
Currently, 10.5% of the world's population has diabetes, yet nearly half remain undiagnosed. Additionally, 10.6% have impaired glucose tolerance and 6.2% have impaired fasting glucose, placing them in a pre-diabetic state. The problem is that these pre-diabetic conditions present almost no symptoms and can go undetected for years, ultimately causing patients to miss the critical window for lifestyle interventions or treatment.
"At-risk participants may remain undetected for years, missing the precious window for early intervention."
Existing blood glucose tests (fasting glucose, oral glucose tolerance, HbA1c, random blood glucose measurement) are all invasive, must be administered by professionals in medical facilities, and are often limited to one-time measurements, making regular and frequent testing difficult.
2. Research Objectives and Methods
The objective of this study was to develop an AI solution that non-invasively detects and classifies elevated blood glucose (7.8 mmol/L or above) and assesses diabetes risk based on repeated measurement data.
Study Design and Data Collection
- Location: KK Women's and Children's Hospital, Singapore
- Participants: 500 individuals (mean age 38.73, mean BMI 24.4), 89.8% female
- Measurements: Participants had blood glucose measured simultaneously with a standard glucometer and a wrist-worn PPG wearable before and after consuming a 75g glucose drink
- Glucometer readings were used as the 'ground truth'
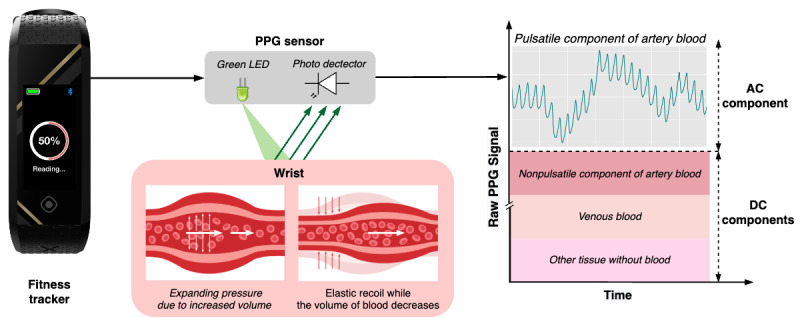
"PPG signals detect changes in light absorption and reflection due to arterial blood flow changes, easily capturing heart rate and vascular health signals from locations such as the wrist."
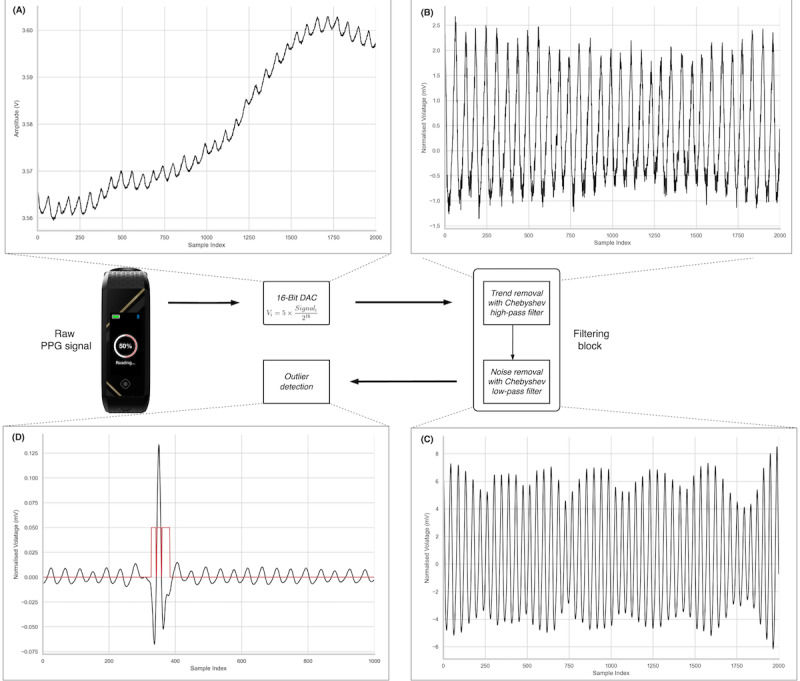
PPG Signal Processing and Feature Extraction
-
After digital-to-analog conversion, noise was removed using a Chebyshev filter
-
Outlier regions were refined through interpolation
-
A total of 248 signal features (heart rate variability, waveform, energy, complexity, wavelet, demographic variables, etc.) were generated
-
Signal preprocessing pipeline:
3. Feature Selection and Machine Learning Model Development
From the high-dimensional features, 12 with the highest predictive power were selected using ensemble feature selection techniques (ANOVA, MI, LASSO, etc.). Notably, demographic factors such as age, BMI, and family history were among them.
| Rank | Feature Name (Example) | Description |
|---|---|---|
| 1 | Welch_hf_rel | Welch power spectrum HF ratio |
| 5 | Age | |
| 9 | BMI | |
| 11 | Family history |
"SHAP analysis reconfirmed that diabetes family history, age, and high BMI are significantly associated with elevated blood glucose risk."
After validating various models using the selected features, SVM (radial kernel) achieved the best performance (accuracy 84.7%, sensitivity 81.1%, specificity 88.3%).
4. Model Interpretation and Practicality
Although deep learning models were briefly considered in the research:
- Insufficient data samples (500 cases)
- Lack of trust and interpretability for 'black box models' in clinical settings
- Ethical considerations
For these reasons, traditional machine learning with explainable AI (XAI: SHAP interpretation) was chosen.
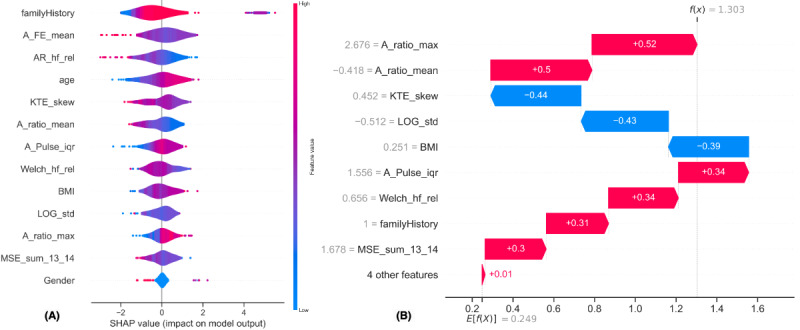
"SHAP values were used to present the degree to which each feature (family history, age, BMI, etc.) positively or negatively influences the prediction outcome."
The ability to clearly interpret how individual features contribute to actual prediction outcomes adds trust in clinical settings.
5. Repeated Measurements and Improved Diagnostic Precision
In general, diagnostic tests struggle to simultaneously satisfy both sensitivity (disease detection) and specificity (distinguishing normal cases). This study demonstrated that repeated measurement values can be combined using 'AND' (both abnormal = high risk) or 'OR' (either abnormal = risk flagged) rules, allowing appropriate combination depending on the desired goal (higher sensitivity vs. higher specificity).
6. Discussion and Limitations
The impact of this research lies in empirically demonstrating the feasibility of non-invasive, low-cost, highly scalable early diabetes screening. If such a solution is commercialized, it could comprehensively identify at an early stage:
- Populations with limited healthcare access
- Regions where regular check-ups are difficult
- Groups requiring lifestyle changes
Furthermore, it could become an innovative tool for repeated self-management and monitoring not only for diabetes patients but also for high-risk groups.
"The non-invasive, device-agnostic solution (BGEM) enables repeated blood glucose monitoring and individualized intervention guidance with minimal disruption."
Limitations and Future Directions
- Fasting glucose was not measured; gender imbalance (predominantly female sample); no long-term follow-up data
- Further expansion to more diverse ages/genders and actual diabetes/pre-diabetes patients is needed
- Independent external validation in separate cohorts is essential
7. Conclusion
This study demonstrated, with a high accuracy of 84.7%, that combining features extracted from wearable PPG signals with a machine learning (SVM) model can non-invasively detect elevated blood glucose and assess diabetes risk. Compared to prior small-scale, controlled-environment studies, sample size and validation performance in real-world settings were significantly improved, suggesting that this approach can evolve into an accurate, low-cost population-level diabetes screening and early management tool.
Closing
This paper supports the strong potential for the convergence of wearable, AI, and signal processing technologies in healthcare to revolutionize early diabetes screening and population health management. While limitations and additional research challenges remain, it inspires hope for a future that offers convenient and effective blood glucose management to a broader range of people.