This paper investigates the molecular and cellular mechanisms behind the rapid-acting antidepressant treatments ketamine and electroconvulsive therapy (ECT). The researchers discovered that adenosine signaling plays a central role in both treatments. Specifically, activation of adenosine A1 and A2A receptors in the mPFC (medial prefrontal cortex) is essential, and the study also presents new ketamine derivatives with fewer side effects and superior efficacy, as well as non-invasive depression treatment strategies leveraging this mechanism.
1. Introduction: Exploring the Mechanism of Fast-Acting Antidepressant Treatment
Numerous clinical and preclinical studies have confirmed that even a single low dose of ketamine produces powerful and sustained antidepressant effects within hours. Thanks to this effect, active research has explored "how ketamine produces such rapid antidepressant effects." Ketamine was initially known as an NMDA receptor antagonist (blocker), but recent work has revealed that it influences a diverse network of neuromodulators.
ECT is also a non-pharmacological treatment that produces very rapid effects in depression. While it has been shown to modulate various neuromodulators, its exact mechanism has not yet been fully elucidated.
This study focused on identifying a common, central neuromodulator across both treatments. Among several candidates, brain adenosine emerged as the most promising.
"Both treatments (ketamine and ECT) must alter neuromodulator concentrations in the brain, and the activation or inhibition of signaling pathways must be able to reproduce or abolish the respective antidepressant effects for the substance to be considered centrally involved."
2. Discovery of Adenosine Surge in the mPFC Following Ketamine Administration
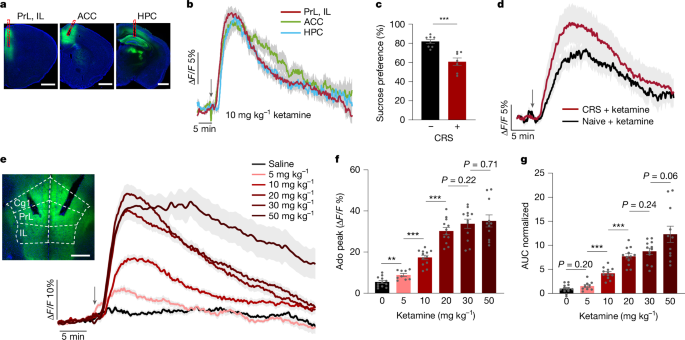
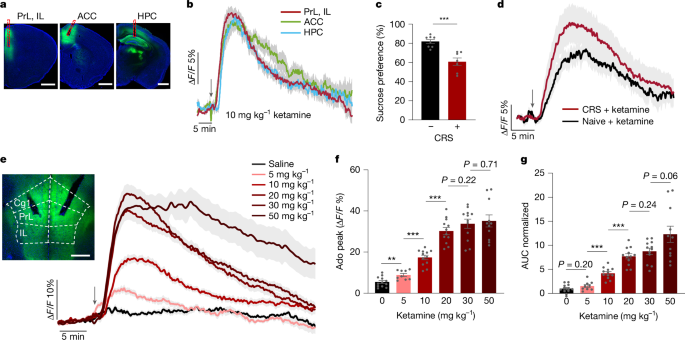
Using a genetically engineered adenosine sensor (GRABAdo1.0), the research team monitored real-time adenosine concentration changes in the mouse brain (particularly the mPFC and hippocampus (HPC)).
After low-dose ketamine injection, adenosine increased rapidly and robustly in the mPFC and hippocampus, with sustained duration. Notably, in depression animal models (CRS), the same adenosine surge was reproducibly triggered with each ketamine dose, just as in normal mice.
Higher ketamine doses increased both the magnitude and duration of the adenosine rise, though at very high doses the signal magnitude plateaued while duration continued to extend.
"Ketamine does not directly activate the adenosine sensor; the effect is confirmed to be endogenous adenosine release."
Furthermore, ketamine metabolites (norketamine, HNK, etc.) did not produce this adenosine surge on their own, while inhibiting the enzyme involved in ketamine metabolism (CYP3A4) enhanced the effect -- establishing that the parent drug ketamine directly induces adenosine release.
3. Proof That Adenosine Signaling Is Essential for Ketamine's Antidepressant Effect
Ketamine's antidepressant effect is easily confirmed through behavioral tests (forced swim, sucrose preference, etc.). However, in mice lacking adenosine receptors (A1, A2A), these effects completely disappeared even when ketamine was administered.
In receptor-knockout mice, the adenosine surge still occurred after ketamine administration, but behavioral antidepressant effects were absent -- proving that adenosine signal transduction (receptor activation) is essential.
"Ketamine's antidepressant action cannot be reproduced without adenosine signaling (specifically A1 and A2A receptors). Neurochemical changes alone are insufficient; the effect only appears when those changes are actually transduced through receptors."
Additionally, temporarily blocking adenosine receptors in normal mice also abolished ketamine's antidepressant effect, while directly injecting adenosine into the mPFC or optogenetically inducing adenosine release produced antidepressant effects similar to ketamine.
4. The Critical Role of mPFC Adenosine Signaling
Injecting adenosine (or adenosine receptor agonists) specifically into the mPFC produced antidepressant effects equivalent to systemic administration, but the same approach in the hippocampus had no effect.
Furthermore, using CRISPR-Cas9 to selectively eliminate adenosine receptor genes only in the mPFC abolished the antidepressant effect of systemic ketamine. This means that adenosine signaling in the mPFC is the critical circuit for ketamine's effect.
"Local adenosine activation in the mPFC is sufficient to produce rapid antidepressant effects, and conversely, blocking the adenosine pathway here also blocks the systemic effect."
5. Elucidating the Metabolic Mechanism of Ketamine-Induced Adenosine Efflux
How adenosine gets released outside the cell has been controversial, but according to this study, ketamine reduces the ATP/ADP ratio in mPFC cells (especially excitatory neurons), resulting in intracellular adenosine accumulation followed by release through ENT1/2 transporters.
Blocking the ENT1/2 adenosine efflux pathway suppressed the ketamine-induced adenosine increase, and it was confirmed that ketamine directly modulates TCA cycle metabolism in mitochondria, reducing ATP supply to drive this process.
"Ketamine acts not only as an NMDA receptor antagonist but as a metabolic modulator in mitochondria, ultimately producing antidepressant effects through adenosine signaling."
6. Exploration and Development of Novel Ketamine Derivatives
Using adenosine surge as a functional biomarker, the researchers synthesized and screened 31 ketamine derivatives. This process yielded novel compounds such as deschloroketamine (DCK) and 2C-DCK with superior efficacy and fewer side effects than existing ketamine.
These new drugs induced adenosine surges and antidepressant effects at lower doses than conventional ketamine, with fewer side effects (such as locomotor excitation). Since there was no correlation between NMDAR (glutamate receptor) blocking tendency and adenosine release, the core effect was clearly mediated by adenosine signaling.
"New drugs with strong adenosine responses showed superior antidepressant effects at lower doses than conventional ketamine, with markedly fewer side effects."

7. Connection to ECT and Non-Pharmacological Treatment (aIH) via Adenosine Signaling
Unlike pharmacological treatment, ECT induces epileptic seizures. Similarly, in mice given ECT, an adenosine surge was triggered in the mPFC comparable to ketamine administration, and ECT's antidepressant effect disappeared in mice lacking adenosine receptors.
Additionally, acute intermittent hypoxia (aIH) -- periodically reducing and restoring oxygen levels -- also increased brain adenosine signaling and produced antidepressant effects in the same manner. Again, the effect was absent in animals lacking adenosine receptors, and side effects were minimal.
"Ketamine, ECT, and aIH all require mPFC adenosine signaling as a common denominator for their effects, and this mechanism alone enables effective and safe new therapeutic approaches."
8. General Discussion and Clinical Implications
This study clearly established that the common, central mechanism of fast-acting antidepressants is brain adenosine signaling. Activation of A1 and A2A receptors is an essential component of the effect, and ketamine's action was revealed to be adenosine-mediated metabolic neuromodulation.
On this basis, not only ketamine and ECT but also safe non-pharmacological approaches (aIH) and new drug development strategies targeting adenosine signaling enhancement could become new solutions for depression treatment.
The study also emphasized that lifestyle factors that block adenosine receptors, such as caffeine, may impair these therapeutic effects and warrant attention in clinical applications.
"We have confirmed that adenosine signaling is the common denominator of rapid antidepressant effects, and we have laid the groundwork for developing safer and more effective treatments leveraging this mechanism."
Closing
This paper vividly demonstrates that the core mechanism of fast-acting antidepressant treatment is adenosine signaling. By elucidating how ketamine, ECT, and even intermittent hypoxia all produce antidepressant effects mediated by mPFC adenosine A1/A2A receptors, the research simultaneously presents drug development strategies that go beyond the side effects of existing treatments, along with significant clinical implications.