This paper reveals how the amygdala–hypothalamus–liver (AMG-VMH-Liver) axis rapidly induces metabolic changes during acute stress. Independent of the classical stress systems (HPA axis, adrenal medulla), a newly identified neural circuit orchestrates key physiological changes during stress — including blood glucose elevation and appetite suppression. Repeated stress impairs the function of this pathway, suggesting a link to metabolic disorders such as type 2 diabetes.
1. Stress and Energy Mobilization: What We Already Knew
When an animal faces a threat or stressful situation, the body rapidly mobilizes energy stores to fuel defensive behaviors such as fleeing or freezing. Cardiovascular and muscular responses are activated, and glucose metabolism shifts accordingly. For example:
"The blood glucose response to stress is closely linked to fight-or-flight behavior."
Hyperglycemia also enhances memory and situational judgment, enabling rapid threat assessment. At the same time, competing behaviors such as feeding and exploration are suppressed. This capacity for metabolic emergency adaptation has been evolutionarily conserved, yet despite its importance, our understanding of the brain circuits that coordinate these responses remains incomplete.
2. Discovery of a New Amygdala–Liver Neural Circuit
The researchers identified a novel amygdala–hypothalamus–liver (AMG-VMH-Liver) axis. This axis operates independently of the HPA axis and adrenal medulla, rapidly elevating blood glucose and suppressing appetite during stress. Repeated stress was found to impair this circuit's function, linking it to metabolic disorders such as type 2 diabetes.
3. Stress-Induced Metabolic Changes
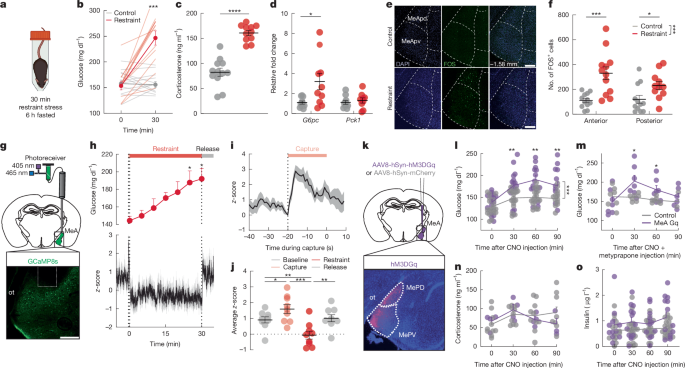
When C57Bl/6 mice were exposed to acute restraint stress or social stress (the scent of a cage occupied by another male), an immediate rise in blood glucose and suppression of appetite were observed.
- Corticosterone, adrenaline, and glucagon levels increased during this process, while insulin and noradrenaline remained unchanged.
- Stress was also confirmed to increase hepatic expression of the glucose production gene G6pc.
- Even stress as brief as five minutes caused rapid increases in blood glucose and stress hormones.
"Restraint stress sharply elevated blood glucose and reduced food intake."
4. Activation and Function of Amygdala (MeA) Neurons
- Stress triggered activation of medial amygdala (MeA) neurons.
- Real-time calcium imaging showed that MeA neurons fire sharply at the moment of capture — before the threat situation fully begins.
- This activation precedes the rise in blood glucose.
- When MeA neurons were artificially excited using DREADD (chemogenetic) techniques:
- Blood glucose rose even without any stress being applied.
- It was confirmed that blood glucose could be elevated without hormonal changes from the classical stress axes (HPA axis, adrenal medulla).
"MeA activation alone was sufficient to raise blood glucose, without any accompanying hormonal changes."
This effect also induced appetite suppression and was not directly associated with anxiety- or fear-related behaviors.
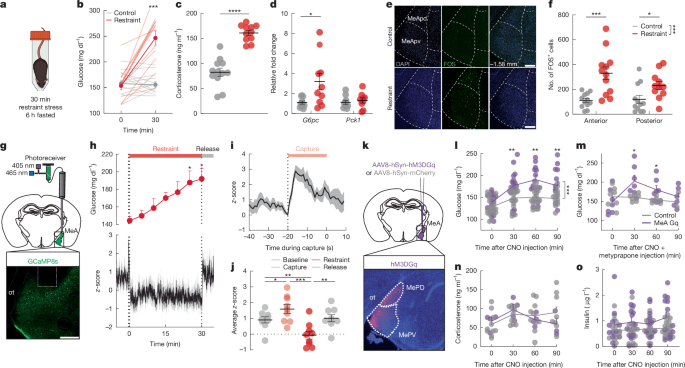
5. Detailed Circuitry: MeA→VMH and MeA→BNST
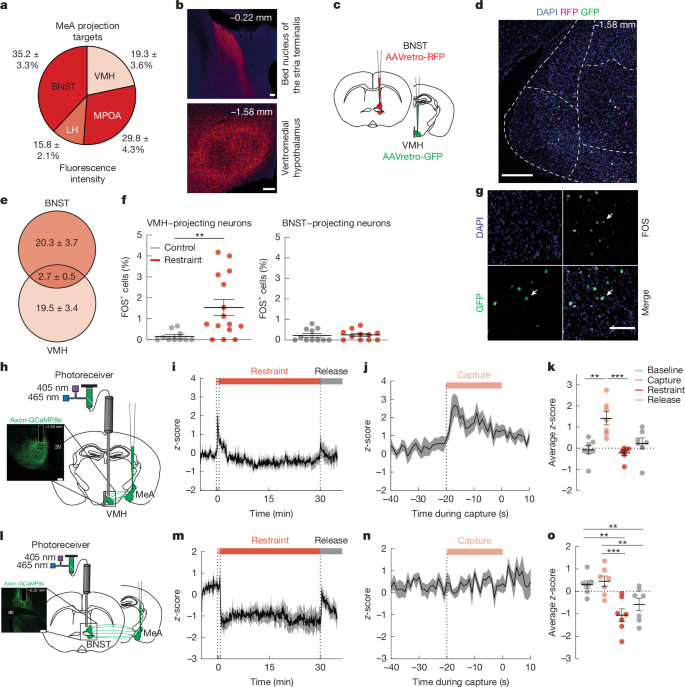
- Neural projections extend from the MeA to the ventromedial hypothalamus (VMH) and the bed nucleus of the stria terminalis (BNST), respectively.
- Under stress, it was confirmed that specifically MeA→VMH neurons are robustly activated and involved in blood glucose elevation.
- The MeA→BNST circuit plays other stress-related roles but has minimal direct involvement in glucose regulation.
"Acute stress primarily activates MeA→VMH neurons."
6. Neuronal Subtypes and Functional Genes
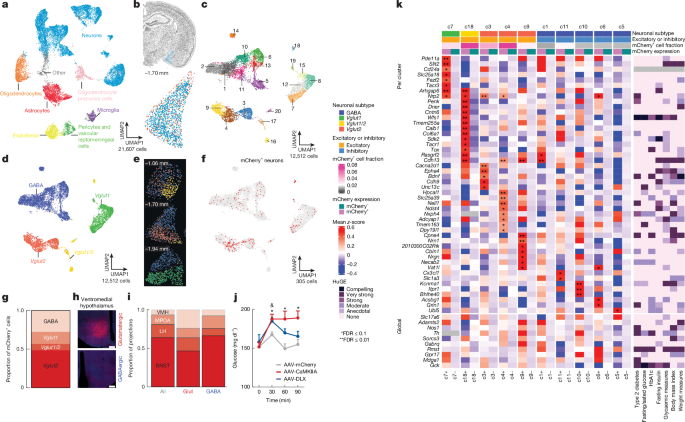
- The MeA→VMH pathway includes both glutamatergic (excitatory) and GABAergic (inhibitory) neurons.
- These neurons show differences in expression of genes associated with hepatic signaling regulation (e.g., G6pc).
- At the genetic level, associations were also found with human changes related to blood glucose, body weight, and diabetes.
"Both excitatory and inhibitory neurons in the MeA→VMH pathway contribute to blood glucose elevation."
7. Direct Hepatic Regulation by the MeA–VMH Circuit
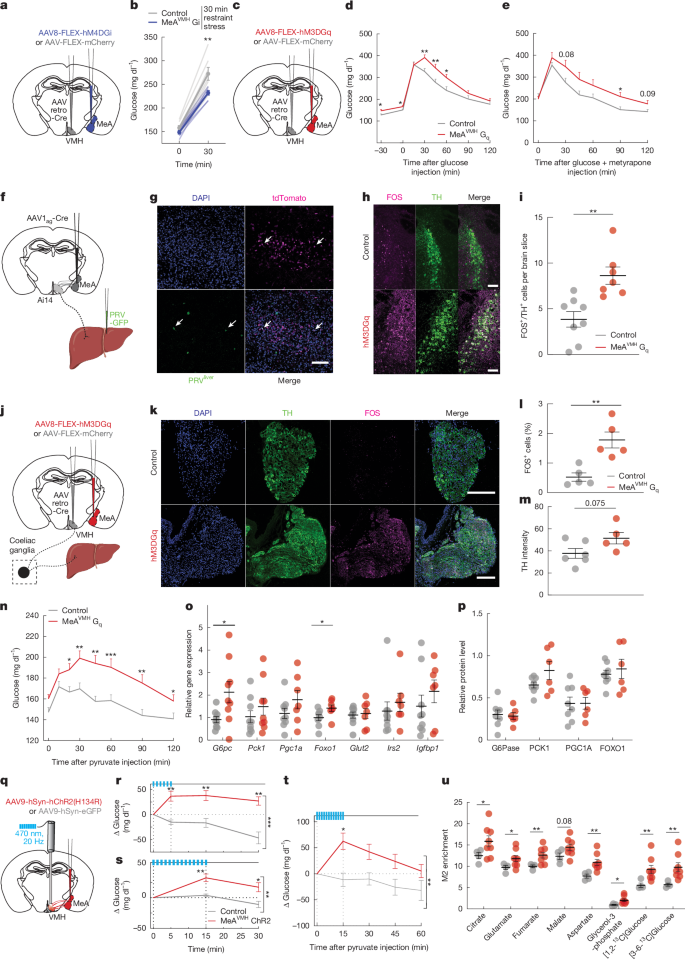
- Blocking MeA→VMH neurons attenuates the hyperglycemic response under stress.
- Conversely, activating these neurons raises blood glucose — an effect independent of "classical hormones" such as insulin, glucagon, and corticosterone.
- A neural pathway extending to the liver was identified: from the VMH, sympathetic outflow promotes hepatic gluconeogenesis peripherally.
- Multi-synaptic connectivity was directly demonstrated using PRV-GFP virus and Cre-based tracing.
- Forced stimulation of this circuit rapidly activates gluconeogenesis-related genes and metabolic processes in the liver.
- This neural circuit creates the flow: increased hepatic glucose production → elevated blood glucose → support for defensive behavior.
"The amygdala–hypothalamus–liver circuit helps the body cope with stress by increasing hepatic glucose production."
8. Repeated Stress Paralyzes the Circuit and Raises Metabolic Disease Risk
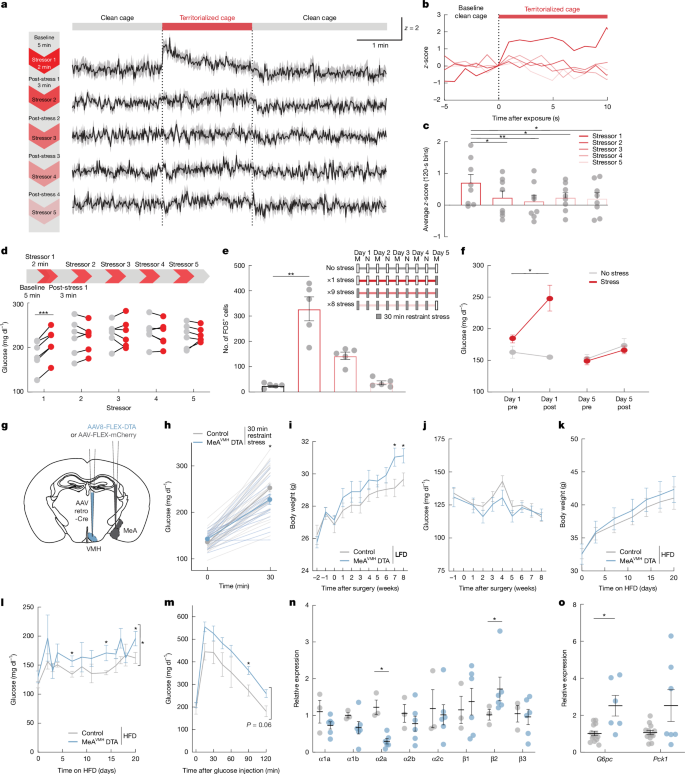
- With repeated stress exposures (e.g., repeated restraint or social stress):
- Blood glucose and neuronal activation rise sharply at first, but progressively diminish with each subsequent exposure (habituation/paralysis phenomenon).
- When MeA→VMH neurons were experimentally ablated:
- Stress-induced blood glucose elevation was reduced, and
- Under metabolic burden (e.g., high-fat diet), glycemic dysregulation, weight gain, and abnormal expression of hepatic metabolic genes were exacerbated.
- In other words, chronic stress or impairment of this circuit is directly associated with increased risk of metabolic disease (obesity, type 2 diabetes, etc.).
"After habituation to repeated stress, paralysis of this circuit drives metabolic dysfunction and weight gain."
9. Conclusion: Stress and Metabolic Disease Through the Brain–Liver Circuit 🌱
This study is significant for identifying the amygdala–hypothalamus–liver circuit — a neural pathway that orchestrates metabolism (particularly blood glucose regulation) during acute stress, operating independently of the classical hormonal axes. Repeated paralysis of this circuit may lead to sustained hyperglycemia and worsening metabolic disease, underscoring the importance of stress management from the perspective of metabolic disease prevention.
"If this mechanism operates in humans as well, aberrant amygdala signaling in chronically stressed individuals could translate to an increased risk of metabolic disease."
Closing Thoughts
This paper offers a fresh neural-circuit-level explanation for why blood glucose rises under stress and why chronic stress may increase the risk of diseases such as diabetes and obesity. It provides an important clue for understanding the brain–body–metabolism connection, and emphasizes the need for an integrated approach to stress management and metabolic disease prevention. 😊