This piece chronicles the remarkable journey of GitLab founder Sid Sijbrandij, who after being diagnosed with cancer in 2022 approached treatment in "Founder Mode" — just as he would scale a company — and ultimately reached remission. Guided by three principles — maximized diagnostics, development of ten-plus personalized therapies, and parallel application of treatments — he overcame the limits of the modern healthcare system and points the way toward the future of cancer care. 🧬
1. A Hunger for Information and the Birth of GitLab 🚀
Sid Sijbrandij is someone who might fairly be called an information maximalist — a person with a deep understanding of information and an exceptional ability to put it to use. In 2003, having studied engineering physics and management science, he worked at a company building personal submarines, absorbed in solving technical problems, but eventually recognized that the industry's growth potential was limited. He then taught himself Ruby on his own and fell deeply into the world of the web and startup culture through Hacker News.
It was there that he discovered GitLab, an open-source collaboration tool created by a Ukrainian developer. He found the Ruby implementation beautiful, and held the conviction that a code collaboration tool's own source code ought to be open source. At the time, unlike GitHub, GitLab was just a repository — users had to download and host the source code themselves — so Sid came up with the idea of building a hosted version called Gitlab.io.
One evening in 2012, while making pancakes with his girlfriend Karen, he posted a link to his beta product on Hacker News. There was no response at first, but soon the comments section exploded with enthusiasm; within three hours, more than 150 people had signed up for early access, giving him the first signal of product-market fit. He immediately emailed GitLab's developer Dmitriy Zaporozhets, who replied, "Great. Please go ahead!" — embodying the open-source spirit.
From there, GitLab gained more and more users. Sid began sponsoring Dmitriy and eventually brought him on as co-founder and CTO. In 2015, they joined Y Combinator (YC) and real growth began.
2. GitLab's Rapid Growth and a Culture of Radical Transparency 🏢
After joining YC, GitLab grew at a remarkable pace. By 2016, it had millions of users, had raised a $20 million Series B, and counted IBM, Macy's, ING, NASA, and VMware among its paying enterprise customers.
At the time, skepticism about remote work was dominant in Silicon Valley, but Sid believed that attracting the best talent required removing geographic constraints. Watching his co-founder Dmitriy collaborate effectively with the team from a small village in Ukraine only strengthened that conviction. GitLab ultimately became one of the world's largest fully remote companies, with more than 2,500 employees and not a single office.
One of GitLab's defining cultural features is radical transparency. The GitLab Handbook — over 3,000 pages long — contains every piece of information about the company and is open not only to employees but to the general public. When an employee has a question, they are expected to check the handbook first; if the answer isn't there, they find it themselves and add it. The GitLab Unfiltered YouTube channel publishes more than 13,000 internal meeting recordings, making even routine information like code reviews transparently available. This unusual way of operating attracted enormous attention from the business community when the COVID-19 pandemic normalized remote work, and Sid became recognized as an expert in managing remote teams.
By 2022, Sid was the CEO of a successful software company, a billionaire, and happily married to Karen, who had been by his side for years. Everything seemed to be going smoothly.
3. An Unexpected Cancer Diagnosis and the Turn to Founder Mode 💔
Then, on November 18, 2022, during a routine workout, he felt a sharp pain in his chest. Similar pains had occurred before, but this one was different, and he ended up in the emergency room. He was sent home after an X-ray, but when a doctor called and he returned to the hospital, he received the shocking news: a 6 cm tumor had been found at his T5 vertebra. 😥
At 45 years old, Sid was diagnosed with osteosarcoma, a rare form of bone cancer. The first goal was tumor removal, and a successful surgery excised the affected vertebral bone, with the spine fused using a titanium frame.
After surgery, he aggressively pursued standard-of-care treatments — stereotactic body radiation therapy (SBRT), intensive chemotherapy, and proton beam therapy. The treatment process was grueling; the chemotherapy was so aggressive that he required four blood transfusions, and he later recalled that it "destroyed him." These treatments left not only temporary side effects but lasting consequences including reduced cardiac function, anemia, and cognitive decline.
Remarkably, amid all this, Sid pursued one additional experimental option. Through his prior investment in Shasqi — an oncology startup founded by José M. Mejía Oneto, a YC classmate from 2015 — he filed an investigational new drug (IND) application with the FDA to apply Shasqi's targeted chemotherapy technology to his own case. With FDA approval, he was able to access it.
4. Relapse and the Beginning of a New Journey 🛤️
Two years of treatment kept the cancer in remission, but a 2024 checkup confirmed local recurrence. Despite surgery, radiation, and the most potent chemotherapy available, the cancer had returned. His medical team had nothing left to offer, and Sid found himself facing what felt like an inescapable death.
"Now it was my job to save myself. Nobody was going to do that for me." With that realization, he reorganized his life. In late 2024, he stepped down as CEO of GitLab and transitioned to Executive Chair, declaring that he would devote his full energy to fighting cancer. He resolved to approach treatment exactly as he would a new venture — in "Founder Mode."
5. Core Principles of Founder Mode and Building a Treatment Team 🛠️
Sid established three core principles for his fight against cancer.
- Maximal Diagnostics: Perform every available diagnostic test as frequently as possible. Just as GitLab operates, the goal was to record and utilize every piece of information, no matter how small.
- Develop 10+ Personalized Therapies: With no standard treatment remaining, he collaborated with corporate and academic researchers to develop new agents informed by detailed information about his specific cancer.
- Parallel Application of Treatments: Rather than using one therapy and moving to the next only after it failed, Sid chose to test multiple treatment hypotheses simultaneously, measuring responses through diagnostics and rapidly progressing to next steps.
These principles diverged sharply from conventional oncology practice. Sid began assembling a team for this journey and engaged a private concierge care service to help design and manage his diagnostic plan.
His new diagnostics regimen fell into five broad categories:
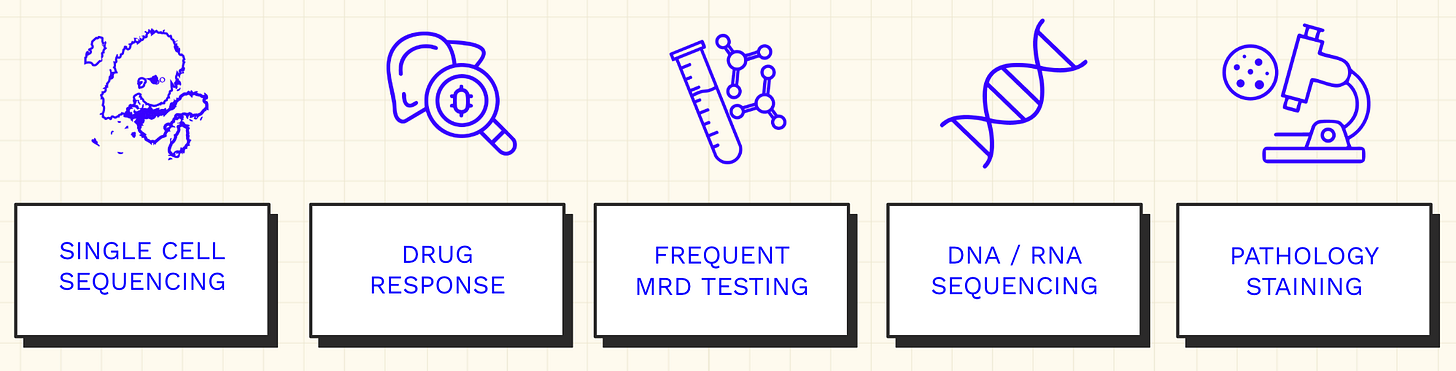
- Single-cell sequencing: Using platforms like 10x Genomics to measure gene expression and analyze T-cell receptors (TCRs) that had infiltrated the tumor, exploring possibilities for targeted immunotherapy.
- Bulk DNA and RNA sequencing: Analyzing the overall genomic profile of cancer cells to characterize the broad mutational landscape.
- Minimal residual disease (MRD) testing: Tracking trace amounts of circulating tumor DNA in the blood through multiple MRD platforms to detect early signs of recurrence.
- Organoid modeling: Collaborating with various labs to create organoids (miniature organs) from his own cancer cells to predict drug responses.
- Histopathological analysis: Using pathology staining on tumor tissue samples to confirm genomic hypotheses.
As part of this effort, Sid recruited Jacob Stern — a board member at 10x Genomics — as a dedicated treatment director. Jacob oversaw the full arc of Sid's care journey, coordinating complex diagnostic and treatment plans, collaborating with hospitals and researchers, and working tirelessly to identify new therapeutic options. He described Sid as "living 30 years in the future."
6. Success with Fibroblast-Targeted Radioligand Therapy 💖
Throughout 2025, Sid and Jacob expanded treatment options at a remarkable pace. A key clue emerged from single-cell data analysis: they confirmed that genes characteristic of fibroblasts were being overexpressed in Sid's cancer cells. Fibroblasts play an important role in wound healing, but cancer cells can exploit them to build a protective barrier within the tumor microenvironment and promote growth.
At the same time, the concierge medical service identified an experimental therapy in development in Germany that directly targets FAP (fibroblast activation protein). Sid said, "I can talk to anyone, go anywhere, any time," and promptly flew to Germany.
The treatment he received there was radioligand therapy — a method that attaches a radioactive isotope to a ligand targeting cancer cells to destroy them precisely. Because the same ligand can be used for both diagnosis (imaging with a "cold" isotope) and treatment (radiation emission with a "hot" isotope), the approach is also called theranostics.
When initial diagnostic imaging clearly revealed Sid's tumor, he proceeded with treatment confidently. He was treated with the isotope lutetium-177, which is also used in other pioneering radiation therapies. After treatment, he was isolated for two days, and over the following two weeks, as the radioisotope was excreted from his body, urine had to be carefully collected and disposed of.

Sid said the radiation therapy was far more tolerable than chemotherapy — it concentrated on the tumor site rather than affecting the entire body systemically. The results were striking: his cancer shrank to the point where surgery was once again possible.
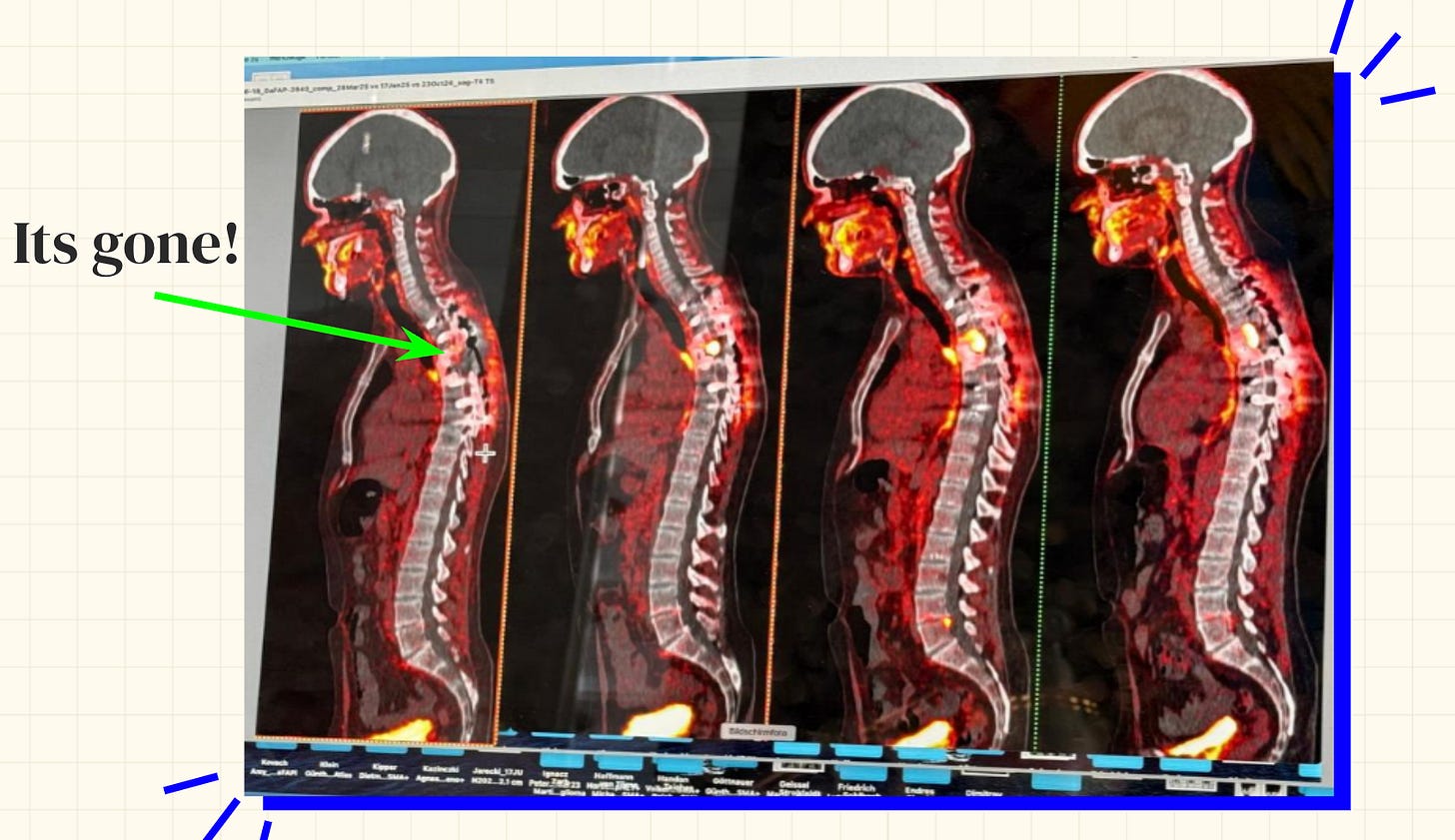
Reanalysis of immune cells within the tumor using fresh tissue obtained at surgery was deeply impressive. When the cancer had relapsed, T cells accounted for only 19% of the infiltrating immune cells. After radioligand therapy followed by surgery, that figure had risen to 89%. This suggests that the immune checkpoint inhibitors, neoantigen peptide vaccines, oncolytic viruses, and radiation therapy Sid had been combining had dramatically activated the immune response against his cancer.
7. Current and Future Treatment Plans: An Unrelenting Pursuit of Cure ✨
Today, Sid's cancer is in remission — no longer detectable, much like Magic Johnson's HIV. But Sid and his team operate under the motto "Stay Paranoid," continuously building out their treatment infrastructure.
The next phase of treatment is a personalized mRNA-based neoantigen vaccine, aimed at sustaining the immune response already induced. Its effectiveness will be evaluated through precise monthly blood tests.
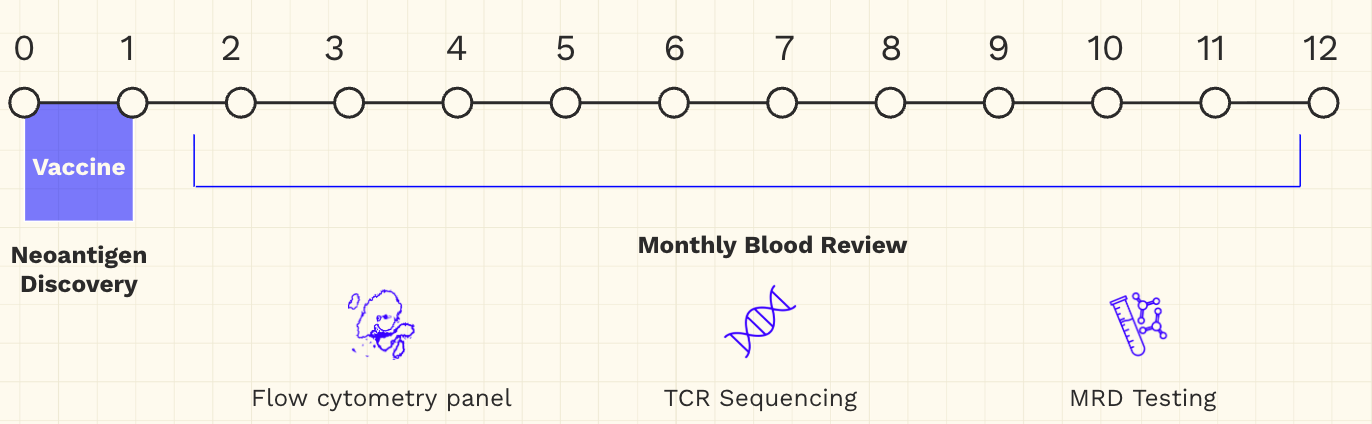
As a contingency, the team is also developing personalized cell-based therapies in collaboration with leading academic research groups. These would equip cells with genetic logic gates that respond to multiple signals to kill cancer cells, informed by even richer diagnostic data. But the ultimate goal is to avoid ever needing this "last resort" by preventing recurrence.
Jacob emphasizes that Sid's case is a "unique example." Sid's extraordinary resources and expertise made this journey possible, but his story stands as important evidence of what future medicine can achieve.
8. Challenges and Potential of the Future Healthcare System 💡
Sid's case clearly illustrates the bottlenecks in modern medicine and where improvement is most needed.
8.1. Resistance from the Hospital System 🏥
Oncologists and hospitals are unaccustomed to patients approaching their care in "Founder Mode." Doctors expect patients to delegate responsibility and follow the standard pathway. Sid described facing "enormous difficulty" at every hospital when trying to access his own tissue samples or pursue non-standard diagnostics. Bureaucracy and liability concerns within institutions created significant resistance to novel approaches.
8.2. The Limits of Genomic Data Utilization 🧬
Since the Human Genome Project, the cost of genomic technology has plummeted, yet whole-genome sequencing remains unintegrated into standard cancer care. Jacob described obtaining sequencing data beyond a standard report as "shockingly difficult." Part of the reason is that sequencing capacity has outpaced the ability to interpret results and translate them into actionable treatment decisions.
8.3. Eroom's Law and Inefficient Drug Development 📉
Cancer is an extraordinarily complex disease that evolves constantly to evade treatment. Compounding this, Eroom's Law — the inverse of Moore's Law — shows that the cost of developing new drugs is rising exponentially. Between 2017 and 2020, the average cost of bringing a new oncology drug to market was $4.4 billion. These enormous costs push pharmaceutical companies to focus only on potential blockbusters, leaving promising experimental compounds with limited market potential to languish. Sid's team often had to hunt down promising experimental drugs at companies on the verge of bankruptcy.
9. The Rise of Personalized Medicine and Regulatory Change 🌟
Nevertheless, Sid's case reveals the promise of tomorrow's personalized medicine.
9.1. Drug Repurposing 💊
Sid used the individual patient expanded access IND (Form 3926) to obtain FDA authorization to access five experimental drugs. Remarkably, the FDA approved his applications within 48 hours, and Sid noted: "The FDA wants me to live." This points to a powerful mechanism for repurposing existing drugs across different cancer types based on diagnostic findings.
9.2. Personalized Drug Development 🧪
Sid collaborated with academic groups and startups to initiate development of several new experimental agents. Recently, CRISPR-based personalized therapies for genetic diseases have been attracting major attention, and the FDA has proposed new regulatory frameworks for such platform technologies. In May 2025, the first personalized CRISPR treatment was administered to an infant with a rare metabolic disorder in Philadelphia, making headlines worldwide.
Similarly, CAR-T therapy — the most significant innovation in cancer treatment in decades — and the personalized neoantigen vaccines Sid is set to receive exemplify precision medicine: therapeutics custom-built for individual patients. Moderna's clinical results in melanoma patients showed that neoantigen vaccines combined with immune checkpoint inhibitors reduced the risk of cancer recurrence or death by nearly half.
These platform technologies are building closed-loop systems that take rich genomic inputs and produce tailored therapeutics, narrowing the gap between measurement and treatment.
10. Scenarios for the Future of Cancer Treatment 🔮
Sid's story is like a glimpse of a future not yet fully realized. His treatment journey required enormous resources and team effort, but in ten or twenty years, a scenario like the following may be possible.
An individual who discovers an anomaly through a consumer cancer diagnostic kit enrolls in a concierge oncology platform. An AI agent analyzes medical records and orders additional tests; a bioinformatics agent generates a deep-analysis report within hours. Diagnosed with early-stage pancreatic cancer, the patient receives — without surgery — immune checkpoint inhibitors (now generic and under $1,000) and an off-the-shelf cancer vaccine calibrated to the tumor microenvironment and antigens, activating the immune system. Personalized radiation therapy then eliminates the tumor, followed by imaging to confirm safety.
No chemotherapy at all. Total treatment cost: $175,000 — far less than the current average for pancreatic cancer treatment (roughly $250,000). The cost is covered by the individual's health savings account and insurer, and everyone comes away satisfied.
Conclusion 🌈
Sid Sijbrandij's cancer journey is more than the success story of one billionaire. It simultaneously reveals the potential of future medicine and the limits of the present system. His "Founder Mode" approach underscores — just as in technology — the importance of maximizing information, developing tailored solutions, and iterating rapidly in the field of medicine as well. His treatments are currently available only to a very few, but as William Gibson said: "The future is already here — it's just not evenly distributed."
Sid's story gives us hope. The effort and resources required today are immense, but if technology continues to advance and the will to change accompanies it, his experience could one day become the universal standard of cancer care. If structural change and innovation in the healthcare system follow, the future of cancer treatment we dream of may arrive sooner than we think. We are grateful for his courage and transparency, and we hope this story brings great value to its readers.
