This article reframes heart rate variability (HRV) not merely as a measure of heartbeat fluctuation, but as a core biomarker of the Brain-Heart Axis (BHA) -- the bidirectional communication network connecting the brain and heart. Drawing on the latest 2025 research, it explains how HRV plays a critical role in predicting and managing not only cardiovascular disease but also neurological and psychiatric disorders, while being closely linked to inflammation regulation and stress response. However, it emphasizes that for widespread clinical adoption, further research is essential to standardize measurements, validate wearable devices, and move beyond mere correlations to establish clear causal relationships.
1. Introduction: Understanding HRV and the Brain-Heart Axis (BHA)
Heart rate variability (HRV) is a measure of how much the time intervals between successive heartbeats vary. This is not simple irregularity but a complex physiological phenomenon that reflects how dynamically our sympathetic nervous system (which increases heart rate) and parasympathetic nervous system (which decreases heart rate) regulate the cardiovascular system. The harmonious interplay between these two nervous systems is essential for maintaining homeostasis.
What is fascinating is that recent research views HRV not merely as a cardiac issue but within the broader framework of the Brain-Heart Axis (BHA). The BHA refers to a bidirectional communication network where the brain's cortex, limbic system, and brainstem regulate cardiac autonomic nerves, while conversely, cardiac signals influence brain activity and emotional regulation.
Disruption of this loop (BHA) due to chronic stress, systemic inflammation, neurodegeneration, or metabolic dysfunction is associated with both primary cardiac diseases (e.g., myocardial infarction, heart failure) and primary neurological/psychiatric disorders (e.g., epilepsy, depression, Alzheimer's disease).
Therefore, a decline or change in HRV may be more than just a signal of cardiac risk -- it could be a warning that the entire system (BHA) is destabilizing. This review aims to help understand HRV's complexity and improve its clinical application.
2. HRV Measurement Techniques and the Importance of Standardization
Methods for measuring HRV are broadly divided into electrocardiography (ECG), used in hospitals, and photoplethysmography (PPG), used in smartwatches and similar devices. While ECG is considered the gold standard, advances in technology have made PPG measurement via smartphone apps and wearable devices increasingly common.
However, caution is warranted. As of 2025, although some studies show high correlation between PPG and ECG, PPG data is vulnerable to motion and light interference artifacts, which can inflate HRV metrics (particularly RMSSD and HF power) by over 30% compared to actual values. Therefore, rigorous validation procedures are needed before trusting wearable device data clinically.
 The table above shows the minimum validation checklist that must be met to treat PPG-based HRV measurements as equivalent to ECG.
The table above shows the minimum validation checklist that must be met to treat PPG-based HRV measurements as equivalent to ECG.
Additionally, HRV results can vary significantly depending on measurement conditions (supine vs. standing, laboratory vs. home), time of day, medications taken, and breathing rate. This is why following standardized reporting checklists like the one below is critically important for research and clinical practice.
 Standardization factors for HRV measurement that should be considered when making clinical decisions.
Standardization factors for HRV measurement that should be considered when making clinical decisions.
Nonlinear parameters also deserve mention. Nonlinear indicators such as fractal analysis and entropy reveal the complexity of heartbeat patterns, but current evidence remains insufficient (Tier 3 level) for definitive clinical conclusions. For now, linear metrics such as SDNN and RMSSD are considered the most reliable (Tier 1) indicators.
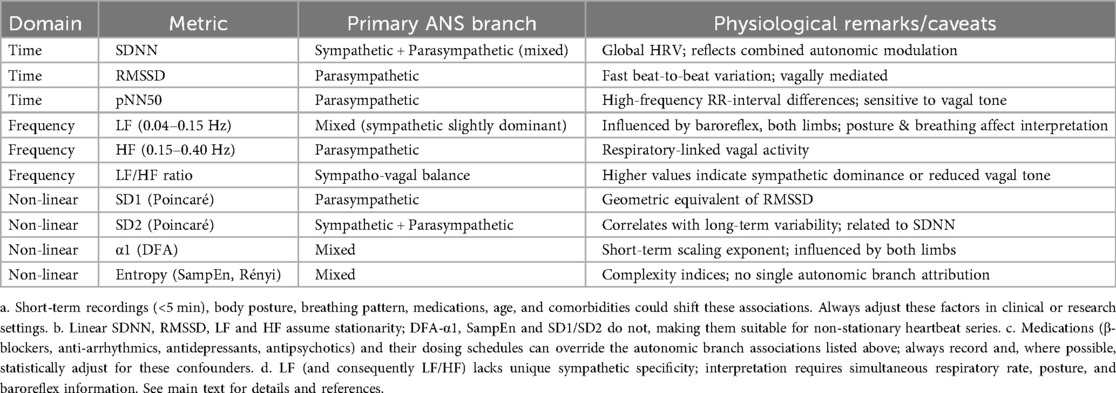 A table summarizing the various HRV components and their corresponding autonomic nervous system branches (sympathetic/parasympathetic) along with physiological caveats.
A table summarizing the various HRV components and their corresponding autonomic nervous system branches (sympathetic/parasympathetic) along with physiological caveats.
3. Mechanisms and Neural Circuits of the Brain-Heart Axis (BHA)
HRV acts as a window into how the brain and heart communicate.
- Brain connectivity: Research shows that individuals with higher resting HRV (RMSSD, etc.) have stronger functional connectivity between the amygdala and prefrontal cortex. This implies superior emotional regulation ability. Conversely, low HRV may indicate difficulty regulating negative emotions during stressful situations.
- Inflammation regulation: The vagus nerve (a key component of the parasympathetic nervous system) plays a crucial role in regulating inflammation. Animal experiments have confirmed that vagus nerve stimulation increases anti-inflammatory cytokines and improves survival rates. In other words, HRV also reflects the body's immune status.
- Transcutaneous auricular vagus nerve stimulation (taVNS): Research has shown that electrically stimulating the auricular branch of the vagus nerve can improve HRV and alter brain wave patterns (frontal theta waves). This opens new possibilities for treating depression and inflammatory diseases.
4. Autonomic Branch-Specific Dysfunction and Clinical Implications
Sympathetic Branch Dysfunction (Cardiovascular Risk)
Low HRV is a powerful signal of elevated cardiovascular disease risk. According to large-scale analyses including the 2025 study by Addleman et al., individuals with resting SDNN below 70ms have a 1.5 to 2.3 times higher risk of major cardiovascular events. Additionally, elevated inflammatory markers (CRP, IL-6) tend to correlate with lower HRV, suggesting that inflammation impairs autonomic nervous system function and serves as a link to cardiovascular disease.
Parasympathetic Branch Dysfunction (Stress and Mental Health)
HRV indicators reflecting parasympathetic activity (RMSSD, HF) are useful for assessing stress resilience and mental health.
- Stress: HRV drops during acute stress, but healthy individuals recover quickly once the stressor is removed. Slow recovery suggests reduced autonomic flexibility.
- Depression: Depressed patients show lower parasympathetic activity than the general population. This may be one reason why depression increases cardiovascular disease risk.
- Biofeedback: Encouragingly, research shows that HRV biofeedback training can restore autonomic balance and improve stress responses.
5. Complex Factors and Conditions: Epilepsy and Obesity
HRV is associated with a wide range of conditions beyond simple cardiac disease.
- Epilepsy: Epilepsy patients, especially those with drug-resistant epilepsy, tend to experience sharp drops in HRV after seizures. This suggests autonomic nervous system dysfunction and may be related to the risk of sudden unexpected death in epilepsy (SUDEP).
- Obesity: Greater waist circumference correlates with lower RMSSD. According to a 2025 study by Wiley et al., low HRV is an independent predictor of future lipid accumulation and cardiometabolic risk. Interestingly, weight loss and lifestyle improvements (exercise, meditation) can improve HRV.
The tables below summarize the various diseases in which HRV has been studied and the specific HRV parameters examined in each condition.
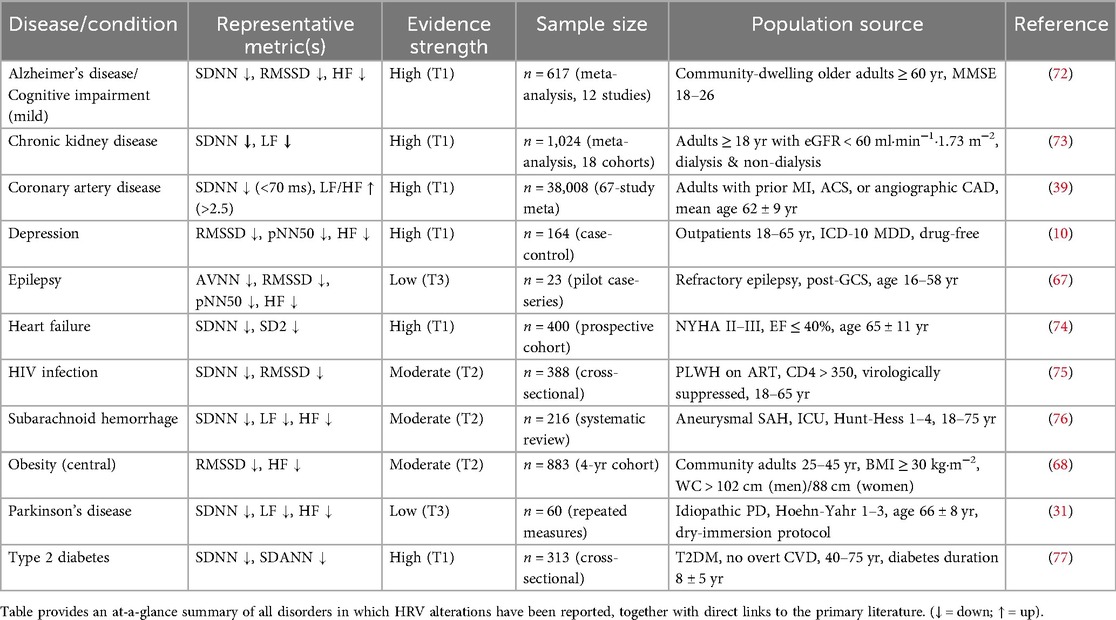 A list of diseases and conditions in which HRV has been studied.
A list of diseases and conditions in which HRV has been studied.
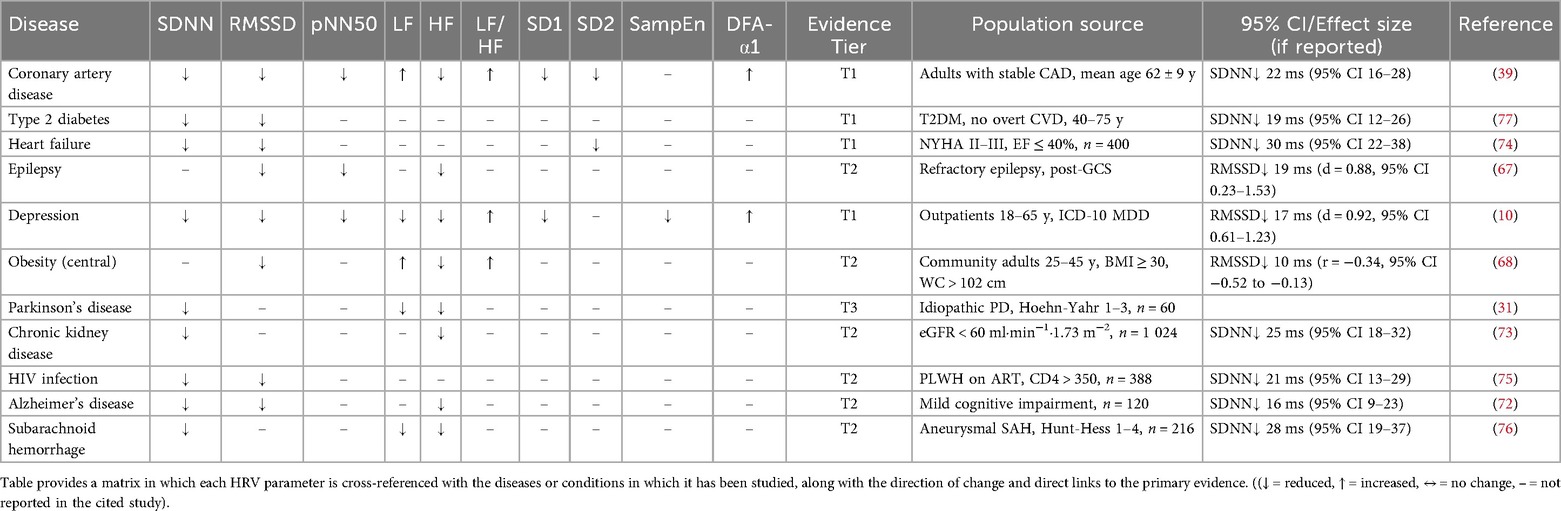 The HRV parameters primarily studied in each disease or condition.
The HRV parameters primarily studied in each disease or condition.
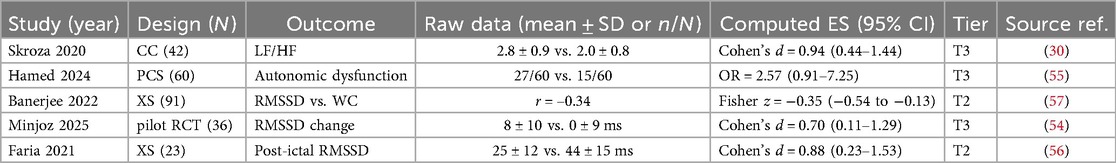 A table computing effect sizes and 95% confidence intervals for studies that did not provide them in the original text.
A table computing effect sizes and 95% confidence intervals for studies that did not provide them in the original text.
6. Current State and Future Outlook of HRV Research
Disease Prediction and Personalized Treatment
HRV is already proving useful in predicting prognosis in heart failure patients and alerting to arrhythmia risk in acute myocardial infarction patients. It also holds great potential for monitoring patients with non-cardiovascular conditions such as Parkinson's disease and dengue fever.
Individualized Medicine
Because each patient has unique genetic backgrounds and physiological responses, HRV can help deliver treatments tailored to the individual. For example, some people show marked HRV improvement from vacation or wellness programs, while others do not. Understanding these individual differences enables more effective health management strategies.
Advances in Artificial Intelligence (AI) and Technology
The proliferation of wearable devices now allows the collection of vast amounts of HRV data from daily life. Combining this with AI and machine learning will greatly increase the accuracy of predicting cardiovascular events and detecting early disease signs. Of course, data quality control and algorithm validation remain essential throughout this process.
Remaining Challenge: Moving from Correlation to Causation
Most current HRV research remains at the level of correlations -- "people with condition A tend to have lower HRV." To use HRV as a genuine clinical biomarker, genetic studies and long-term follow-up observations are needed to determine whether HRV changes actually cause disease.
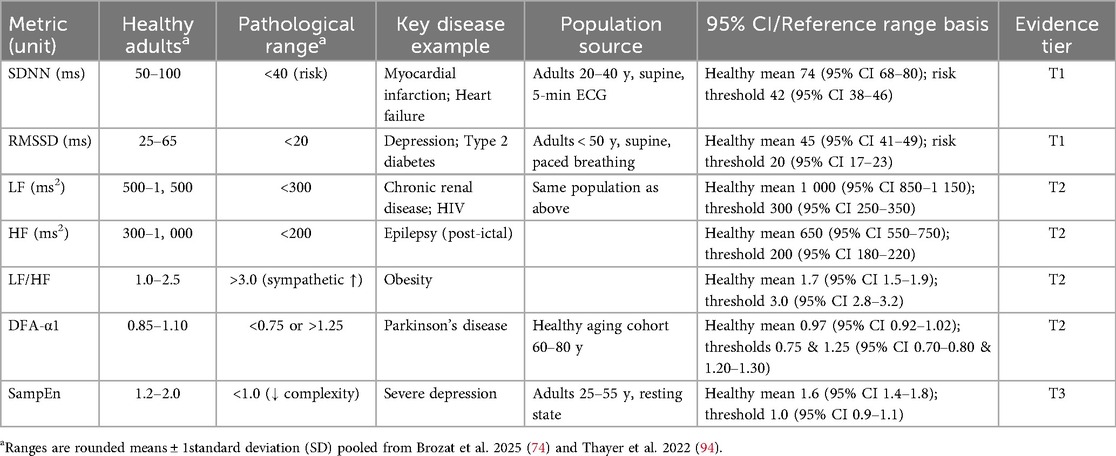 Commonly reported short-term (5-minute, supine) HRV reference ranges for healthy adults and individuals with various conditions.
Commonly reported short-term (5-minute, supine) HRV reference ranges for healthy adults and individuals with various conditions.
Conclusion: Potential and Challenges as a Biomarker
Through this review, we have confirmed that HRV is far more than a simple cardiac indicator -- it is a critical measure of brain-heart connectivity (BHA). The reduction in HRV observed across cardiovascular disease, obesity, depression, epilepsy, and other conditions is a shared signal that the body's autonomic regulation system is breaking down.
The fact that HRV reflects the disruption of this loop (connecting brain and heart) repositions HRV from a peripheral cardiovascular measurement to a central nervous system-cardiovascular coupling biomarker.
With standardized measurement methods and rigorous research to back it up, HRV has the potential to become a powerful tool for early disease detection and treatment monitoring. However, it must not be forgotten that for now, HRV should be interpreted cautiously when making clinical decisions outside of research contexts.
