This study proposes a new framework for predicting insulin resistance (IR) using data from wearable devices, demographic information, and routine blood biomarkers. In particular, it uses a wearable foundation model (WFM) to substantially improve prediction accuracy, and develops an LLM agent called IR Agent that provides personalized health advice based on prediction results. Together, these methods open new possibilities for diabetes prevention and management.
1. Understanding Insulin Resistance and the Importance of Early Detection
As of 2026, the global number of people with diabetes is 537 million, and it is expected to rise to 643 million by 2030. About 90% of cases are type 2 diabetes (T2D), which is closely related to lifestyle. In healthy people, insulin secreted by pancreatic beta cells helps cells absorb glucose from the blood and regulate blood sugar. But most people with T2D develop insulin resistance (IR), meaning they must produce more insulin to achieve the same blood-glucose-lowering effect. Over time, pancreatic beta cells can no longer produce enough insulin, leading to relative insulin deficiency and rising blood glucose.
IR affects roughly 20-40% of the general population and can arise from excess body weight, especially visceral fat, lack of physical activity, and genetic factors. Long-term IR greatly increases the risk of prediabetes, overt T2D, metabolic dysfunction-associated steatotic liver disease, and cardiovascular disease. That makes early detection of IR extremely important. If IR is identified early, personalized lifestyle interventions such as weight loss, regular exercise, and a healthy diet may improve or even reverse it.
There are several ways to assess IR, but most are difficult to use routinely and often lack sensitivity for early-stage IR. For example, the hyperinsulinaemic euglycaemic clamp is the gold standard for measuring IR, but it is possible only in research facilities and is expensive and time-consuming. HOMA-IR is cheaper and faster, but still requires a clinical laboratory visit. Biometric signals from smartwatches may help predict IR, since studies have linked higher resting heart rate and lower heart-rate variability with IR.
To overcome these limitations, this study presents a method for predicting IR using signals from consumer smartwatches, demographic information, and routine blood biomarkers. The approach has the potential to be applied to millions of people and to enable broad identification of IR.
2. Study Design and Participant Characteristics
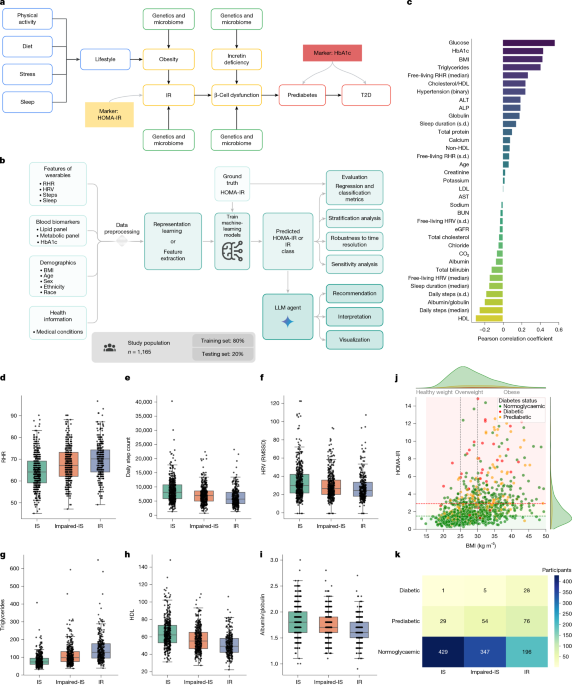
The research team designed the Wearables for Metabolic Health (WEAR-ME) study and recruited adult participants in the United States. Participants collected data from wearable devices such as Fitbit and Google Pixel Watch through the Google Health Studies app, completed questionnaires, and received blood tests through Quest Diagnostics.
The reference value for quantifying IR was the HOMA-IR index. HOMA-IR is calculated as fasting insulin multiplied by fasting glucose, divided by 405. Participants were classified by HOMA-IR as follows:
- IR (Insulin Resistance): HOMA-IR > 2.9
- IS (Insulin Sensitivity): HOMA-IR < 1.5
- Impaired-IS (Impaired Insulin Sensitivity): 1.5 <= HOMA-IR <= 2.9
In the final analysis, 1,165 participants were included: 459 in the IS group, 406 in the Impaired-IS group, and 300 in the IR group. Participants in the IR group had higher rates of diabetes, cardiovascular disease, hyperlipidemia, and hypertension.
The team built a deep learning framework for IR prediction. The framework takes wearable data, blood biomarkers, demographic information, and other inputs, predicts HOMA-IR values, and classifies IR based on those values. In particular, it uses a masked autoencoder (MAE) to extract embeddings from time-series data and feeds those embeddings into multiple tree-based models to predict HOMA-IR.
3. Associations Between IR, Lifestyle, and Blood Biomarkers
The study found significant correlations between HOMA-IR and major lifestyle factors and blood biomarkers.
- Positive correlations:
- Fasting glucose (r = 0.57, P < 0.001)
- BMI (r = 0.43, P < 0.001)
- HbA1c (r = 0.45, P < 0.001)
- Triglycerides (r = 0.40, P < 0.001)
- Resting heart rate (RHR) (r = 0.27, P < 0.001)
- Negative correlations:
- High-density lipoprotein (HDL) cholesterol (r = -0.30, P < 0.001)
- Daily step count (r = -0.25, P < 0.001)
- Albumin/globulin ratio (r = -0.18, P < 0.001)
- Heart-rate variability (HRV) (r = -0.14, P < 0.001)
These results suggest that HOMA-IR can be predicted from wearable devices and blood biomarkers. CRP levels were also significantly higher in the IR group than in the IS group, at 2.8 mg/dL versus 0.6 mg/dL (P < 0.001).
The relationship between obesity and IR was also clear. Among 458 participants with obesity (BMI > 30), 205 people (45%) had insulin resistance. Among 319 participants with normal weight (18.5 < BMI < 25), only 22 people (6.9%) had insulin resistance.
The relationship between IR and diabetes is also notable. Among 34 people with diabetes (HbA1c > 6.5%), 33 people (97%) were classified as IR or Impaired-IS, and only one person was IS. Especially important is the fact that among 972 people whose HbA1c appeared normal, 196 people (20%) had IR. Because this group is at higher risk of developing diabetes, personalized lifestyle intervention may be able to reverse progression toward T2D.
4. Predicting IR with Wearables and Blood Biomarkers
The research team developed a multimodal model that combines wearable data, demographic information, and blood biomarkers to predict IR. The model first predicts HOMA-IR and then classifies IR based on that value.
Using wearable data, demographic information, and routine blood biomarkers together substantially improved prediction accuracy.
- Wearables + demographics only: R² = 0.212
- Adding fasting glucose: R² increased more than twofold to 0.435, accurate identification of IR patients increased by 17%, and false negatives, where IR was misclassified as IS, decreased by 46%
- Best model (wearables + demographics + fasting glucose + lipid panel + metabolic panel): R² = 0.50
This suggests that fasting glucose alone is insufficient, and that other lifestyle factors are very important for estimating HOMA-IR. The model also performed well for IR classification:
- Wearables + demographics only: AUROC = 0.70, sensitivity = 0.60, specificity = 0.80
- Adding fasting glucose: AUROC = 0.78, sensitivity = 0.73, specificity = 0.84
- Best model (wearables + demographics + routine blood biomarkers): AUROC = 0.80, sensitivity = 0.76, specificity = 0.84
These results show that integrating multiple data sources greatly improves the ability to predict IR.
5. Predicting IR with a Wearable Foundation Model
To use wearable data more effectively, the researchers tested an approach called a wearable foundation model (WFM). A WFM learns complex, high-dimensional feature representations from large amounts of unlabeled wearable data. Such foundation models are useful for learning robust features from complex time-series data.
The WFM was pretrained on 40 million hours of Fitbit user data to extract features from wearable signals at one-minute resolution. Each participant's data was aggregated by day into a single embedding, then reduced in dimensionality and used for IR prediction.
Using WFM-derived feature embeddings further improved IR prediction.
- WFM-based model (WFM embeddings + demographics): AUROC = 0.82, a major improvement over demographics alone at 0.66
- Best WFM-based model (WFM embeddings + demographics + fasting glucose + lipid panel): AUROC = 0.87, a major improvement over the same model without wearable data at 0.78
In SHAP analysis, the contribution of wearable data in the WFM-based model was 82%, much higher than the 43% contribution in the conventional machine-learning model. This means the WFM effectively captures complex dynamic interactions relevant to IR, such as daily activity, sleep, and physiological rhythms.
6. Independent Cohort Validation and IR Agent Development
To test generalizability, the researchers evaluated the model in an independent validation cohort. This cohort included 72 participants and, as in the initial cohort, used HOMA-IR as the reference value for IR.
The validation results again showed that wearable data provides substantial value for predicting IR even in previously unseen data:
- WFM-based model (WFM embeddings + demographics): AUROC = 0.75, improved over demographics alone at 0.66
- Best WFM-based model (WFM embeddings + demographics + fasting glucose + lipid panel): AUROC = 0.88, a major improvement over the same model without wearable data at 0.76
These results show that a WFM can fully leverage the potential of wearable sensor data to provide a robust and generalizable method for metabolic health assessment.
The researchers then developed IR Agent, a large language model agent that uses predicted IR information to provide personalized health advice. IR Agent is based on Gemini 2.0 Flash and uses the ReAct (Reasoning and Acting) framework. Alongside language understanding, it interacts with external tools such as web search, calculators, and the IR prediction model. It dynamically plans answers to user questions and grounds them in actual data and verifiable calculations.
To evaluate IR Agent, five endocrinologists conducted blinded assessments. They found that IR Agent provided answers that were more comprehensive (80%), trustworthy (92%), and personalized (73.3%) than those from a general LLM. Its responses were also rated highly for factual accuracy (79%) and safety (96%).
"IR Agent provided more comprehensive, trustworthy, and personalized answers than a general LLM."
7. Conclusion
This study presents the first deployable end-to-end model for predicting insulin resistance using wearable devices, demographic information, and routine blood biomarkers. In particular, the wearable foundation model achieved much higher prediction accuracy than conventional simple aggregation methods, and that advantage held in an independent validation cohort.
Today, 26% of the U.S. population owns a smartwatch, and 15% receives an annual routine checkup. In this context, an IR prediction model that combines wearables and blood biomarkers could provide a scalable solution for early identification of IR in millions of people. Given that existing IR diagnostic methods are difficult to use routinely because of cost and time, this approach has the potential to greatly expand access to metabolic health screening.
The team also developed IR Agent, an LLM-based assistant that uses the IR prediction model to provide personalized health information and advice. Endocrinologist evaluations recognized its usefulness and safety, suggesting that AI-based health assistants like this may play an important role in early prevention and management of chronic diseases such as diabetes.
Of course, the study has limitations. For example, only 25% of participants had complete data, and important IR-related data such as genetic factors and the microbiome were not included. Still, as wearable adoption spreads and research continues, these limitations may be overcome, enabling more precise and personalized IR prediction and management.
Ultimately, the study shows the innovative potential of wearable technology and AI for early detection and management of insulin resistance, offering new hope in the fight against diabetes.