This article provides an in-depth exploration of how heart rate variability (HRV) reflects autonomic nervous system regulation of the cardiovascular system and can be utilized for predicting and treating various diseases. In particular, it redefines HRV as an integrative biomarker of the Brain-Heart Axis (BHA), emphasizing its importance not only in cardiac diseases but also in neurological and psychiatric conditions. The text covers standardization of HRV measurement methods, influencing factors, the significance of linear and nonlinear HRV parameters, and clinical applications and future research directions.
1. What Is Heart Rate Variability (HRV) and Why Does It Matter?
Heart rate variability (HRV) refers to the subtle variations in time intervals between consecutive heartbeats. Beyond simply measuring heart rate, it is a complex physiological phenomenon that reveals how flexibly our autonomic nervous system (ANS) regulates the cardiovascular system. The ANS consists of the sympathetic nervous system, which accelerates heartbeats, and the parasympathetic nervous system, which slows them down, and the balance between these two systems is crucial for maintaining cardiovascular homeostasis.
Recent research suggests that HRV arises not merely from cardiac changes alone but from the interplay of multiple factors including central autonomic networks, intrinsic cardiac activity, baroreflex, respiratory-cardiac coupling, and hormonal changes. Notably, a hypothesis has been proposed that the amplitude of low-frequency HRV (LF-HRV) serves as a reference input for neurocardiovascular centers, which respond by attempting to maintain LF-HRV at a specific level.
HRV research has an exceptionally broad scope of importance and application. Associated with diverse health conditions, it can serve as a valuable biomarker in both clinical and research settings. Studies have shown that low HRV is associated with increased all-cause mortality and cardiac-related mortality.
"A meta-analysis including two individual participant data sets from 32 studies and 38,008 participants showed that low HRV parameter values were significant predictors of higher mortality across diverse ages, sexes, continents, populations, and recording lengths."
Furthermore, HRV biofeedback has shown positive effects in various domains, such as reducing autonomic nervous system responses during anger situations and aiding recovery in coronary artery disease patients. This suggests that HRV has potential as a therapeutic tool that can contribute to health promotion and disease prevention.
1.1 The Importance of the Brain-Heart Axis (BHA)
The Brain-Heart Axis (BHA) refers to the bidirectional neuro-hormonal communication network between the brain and heart. Cortical, limbic, and brainstem centers in the brain regulate the cardiac autonomic nervous system, while conversely, cardiac signals influence brain activity and emotional regulation. When this loop is disrupted by chronic stress, systemic inflammation, neurodegeneration, or metabolic autonomic dysfunction, it can affect not only cardiac diseases (e.g., myocardial infarction, heart failure) but also neurological/psychiatric conditions (e.g., epilepsy, depression, Alzheimer's disease).
Because HRV can non-invasively measure vagal nerve and sympathetic nerve output in real time, it serves as an integrative window revealing the integrity of the BHA. Therefore, changes in HRV may signal not just simple cardiac risk but broader BHA disturbances. In other words, HRV needs to be reinterpreted from a peripheral cardiovascular marker to a central nervous system-cardiovascular coupling biomarker.
In this context, this study aims to comprehensively report on factors affecting HRV measurement, its role in cardiovascular disease, neurology, and lifestyle assessment, as well as its current state and future prospects. The goal is to deepen understanding of HRV's complexity and support clinicians in better utilizing HRV for health monitoring and disease diagnosis in clinical settings.
1.2 Conceptual Framework
This study examines the relationship between BHA integrity and HRV along three axes:
- Measurement axis: Defines which metrics capture which aspects of the BHA.
- Mechanistic axis: Explains how BHA disturbances (stress, inflammation, neurodegeneration) manifest as specific HRV patterns.
- Translational/predictive axis: Explores how HRV-based interventions restore BHA integrity and improve both cardiac and neurological dual outcomes.
Additionally, the following three testable claims will be evaluated:
- Reduced HRV consistently indicates BHA disturbance across cardiac, metabolic, and neuropsychiatric disorders.
- Nonlinear HRV parameters decline first when central autonomic network integrity is lost.
- HRV biofeedback/lifestyle interventions that increase vagal tone simultaneously improve cardiac and neurological outcomes through BHA restoration.
2. Introduction to HRV and the Autonomic Nervous System (ANS)
HRV arises from the dynamic interplay of sympathetic and parasympathetic nerves that continuously fine-tune the pacemaker activity of the cardiac sinoatrial node. Therefore, all HRV metrics can ultimately be mapped to one or both autonomic branches.
- The sympathetic nervous system releases noradrenaline to accelerate heart rate and shift spectral power into the low-frequency (LF) band (approximately 0.04-0.15 Hz). Sympathetic hyperactivity observed in heart failure, acute myocardial infarction, and psychosocial stress manifests as decreased standard deviation of normal-to-normal intervals (SDNN), elevated LF/HF ratio, and blunted nonlinear complexity.
- The parasympathetic nervous system releases acetylcholine to induce bradycardia and high-frequency (HF) oscillations (approximately 0.15-0.40 Hz). Parasympathetic withdrawal seen after major depression and epileptic seizures is measured through decreased root mean square of successive NN interval differences (RMSSD), decreased percentage of NN intervals differing by more than 50ms (pNN50), and decreased high-frequency (HF) power.
Most real-world recordings reflect simultaneous sympathetic and parasympathetic modulation. Global metrics such as SDNN and SD2 (long axis of the Poincare plot) capture these combined influences, while nonlinear metrics such as SD2/SD1 ratio and DFA-alpha1 quantify system complexity. With aging, these mixed signals progressively weaken, highlighting HRV's role as a biomarker of age-related decline in autonomic function.
2.1 Measurement Techniques and Standardization
Multiple techniques are used for HRV measurement, and standardization is critically important for obtaining accurate and comparable results. Electrocardiography (ECG) is the gold standard method for obtaining R-R intervals, from which HRV indices are calculated. However, with technological advances, smartphone apps using photoplethysmography (PPG) are also being used for HRV measurement.
For example, in 2022, Moya-Ramon et al. validated Elite HRV (chest strap ECG) and Welltory (PPG) against 12-lead ECG in 30 elite cyclists. These apps showed no differences from ECG in supine and seated positions, with very strong to near-perfect correlations of r = 0.77-0.94. However, commercial smartphone/PPG apps often lack raw data transparency, making manual ectopic beat editing and arrhythmia screening difficult. This can cause artifacts to inflate RMSSD or HF power by over 30%, producing variability that appears "high" but is clinically meaningless.
"Commercial smartphone/PPG apps often lack raw data transparency, making manual ectopic beat editing and arrhythmia screening difficult. As a result, artifacts can inflate RMSSD or HF power by over 30%, producing artificially 'high' but clinically meaningless variability."
PPG-based interbeat intervals are inherently affected by pulse transit time variability (PTTV) and arterial compliance changes due to posture, ambient temperature, and exercise intensity. During active standing or walking, PTTV introduces plus/minus 20-40ms beat-to-beat variance of non-neurological origin, which can cause systematic overestimation of LF power and underestimation of vagal indices (RMSSD, HF). Additionally, most consumer wearables buffer optical data at 20-50Hz, where suboptimal peak detection algorithms can introduce 5-15ms timing errors that increase SDNN by 5-10% and corrupt entropy measurements. Therefore, PPG-based HRV cannot be equated with ECG metrics and requires rigorous validation protocols (see Table 1).
 Table 1. Validation checklist.
Table 1. Validation checklist.
The standardization of HRV measurement is essential due to the numerous factors affecting HRV values. According to a 2025 study by Besson et al., which investigated the reliability of short-term HRV measurements across different environments (home vs. laboratory) and postures (supine vs. standing), the environment significantly influenced standing-posture HRV. Home measurements showed slightly lower variance than laboratory settings. This highlights the need for controlled conditions and consistent protocols in HRV monitoring and interpretation, which is important for ensuring accuracy and comparability of results across various research and clinical applications (see Table 2).
 Table 2. Clinical decisions.
Table 2. Clinical decisions.
Reporting Checklist (adapted from Laborde et al. 2017 and Task Force 1996)
- Recording duration: Short-term >= 5 minutes (ultra-short <= 1 minute for RMSSD only); prognostic indices require 24 hours.
- ECG sampling frequency: >= 256 Hz recommended; <= 500 Hz acceptable with hardware limitations.
- Artifact/ectopic beat detection: Automated algorithm + manual inspection; ectopic beats >= 5% -> exclude segment or report pre/post-editing values.
- Interpolation: Gaps of 3 beats or fewer use cubic spline or Lomb-Scargle; more than 3 consecutive gaps -> discard segment.
- Posture: Supine, seated, or standing; specify habituation period.
- Timing: Report recording time and whether fasting/exercise occurred within the preceding 3 hours.
- Medication log: Record drug type, dose, time of last dose relative to recording; include sympatholytics, anticholinergics, beta-blockers, etc.
- Environmental conditions: Quiet, temperature 22-24 degrees C; home vs. laboratory setting.
- Data sharing: Provide de-identified IBI series and codebook for reanalysis (e.g., PhysioNet-compatible format).
2.2 Factors Affecting HRV
HRV is influenced by multiple factors including age, sex, disease state, and lifestyle. Exercise, postural changes, circadian rhythms of cortisol secretion, and sex steroid fluctuations also modify HRV through dynamic autonomic nervous system actions and direct electrophysiological effects. For example, in patients with atrial septal defect (ASD), age, sex, defect diameter, heart rate, and diabetes were associated with HRV indices. In a study of 154 ASD patients who underwent percutaneous closure, SDNN and SDANN significantly increased after closure, and these indices showed clear correlations with right ventricular systolic pressure.
Breathing patterns also significantly affect HRV. Spontaneous breathing at 15 breaths per minute (0.25Hz) increases HF power and decreases LF, while breathing at 0.1Hz synchronizes with LF and can artificially elevate the LF/HF ratio even without changes in sympathetic output.
Mental health status also affects HRV. In patients with schizophrenia and major depressive disorder, age-related HRV changes were observed, with adult patients showing decreased time-domain and nonlinear HRV compared to adolescents. Additionally, female patients had lower time-domain HRV, LF/HF, and SD2 than males. Stress and negative affect also influence HRV; in studies of patients with functional somatic syndrome, high negative affect and comorbid depression were associated with HRV changes.
2.3 Effects of Medications on HRV
Autonomic modulation by medications is a major source of uncontrolled variability.
- Beta-blockers, non-dihydropyridine calcium channel blockers, and centrally acting sympatholytics generally increase RMSSD and HF power.
- Conversely, tricyclic antidepressants, anticholinergics, and some antipsychotics decrease vagal indices.
Therefore, when using baseline HRV for risk stratification, complete medication history, dosage, and timing of administration are essential. Failure to account for these medications can cause SDNN to fluctuate by more than 20ms and LF/HF by more than 1 unit.
2.4 Nonlinear HRV Parameters
Beyond linear metrics, nonlinear HRV analysis captures the complexity and irregularity of cardiac rhythm. Poincare plot-based indices (SD1, SD2, SD2/SD1) and entropy-based measurements (ApEn, SampEn) provide additional insights into sympatho-vagal balance, while detrended fluctuation analysis (DFA)-alpha1/alpha2 and correlation dimension (D2) quantify fractal properties and system complexity that are often blunted in autonomic dysfunction.
- Poincare Plot Geometry
- SD1 (short-axis variance): Dominated by parasympathetic modulation, showing strong correlation with RMSSD.
- SD2 (long-axis variance): Reflects combined sympathetic and parasympathetic influences, analogous to SDNN and LF power.
- SD2/SD1 ratio: Provides a geometric analog of the LF/HF ratio and tracks sympatho-vagal balance shifts.
- Entropy Measures
- Approximate Entropy (ApEn) and Sample Entropy (SampEn): Quantify signal regularity. Both indices increase during mental stress and are related to decreased autonomic complexity rather than pure vagal withdrawal.
- Detrended Fluctuation Analysis (DFA)
- alpha1 (short-term scaling exponent): Modulated by both autonomic branches.
- alpha2 (long-term exponent): Increases with relative shifts toward sympathetic dominance and decreased vagal activity.
- Correlation Dimension (D2): Low D2 indicates loss of nonlinear complexity, associated with impaired parasympathetic modulation in HIV-positive patients receiving antiretroviral therapy.
Nonlinear HRV parameters have emerged as important tools for understanding the complex dynamics of cardiac autonomic regulation across various physiological and psychological conditions. These parameters provide insights into the intricate interactions between the sympathetic and parasympathetic nervous systems that go beyond traditional linear metrics.
Research shows that nonlinear HRV parameters are sensitive indicators of mental and physical stress. For example, studies comparing HRV during paced breathing and mental arithmetic tasks found significant differences in nonlinear parameters, highlighting their usefulness in distinguishing between relaxation and stress states. Research has also shown that psychological workload significantly reduces HRV complexity.
Regarding exercise, nonlinear HRV parameters have been shown to reflect the body's adaptive responses. The nonlinear dynamics of HRV measured through DFA during cycling show decreased complexity with increasing exercise intensity, suggesting a shift from autonomic to non-autonomic regulation at higher intensities.
In clinical settings, nonlinear HRV parameters also provide insights into disease states and treatment interventions. In Parkinson's disease patients, nonlinear HRV metrics have been used to indicate compensatory mechanisms of cardiovascular regulation despite neurodegeneration. Additionally, in treating secondary anemia in frail elderly patients, nonlinear HRV analysis has been used as a predictor of cardiovascular tolerability to assess treatment safety.
All these parameters are included in Table 3 along with their corresponding ANS branches and physiological caveats.
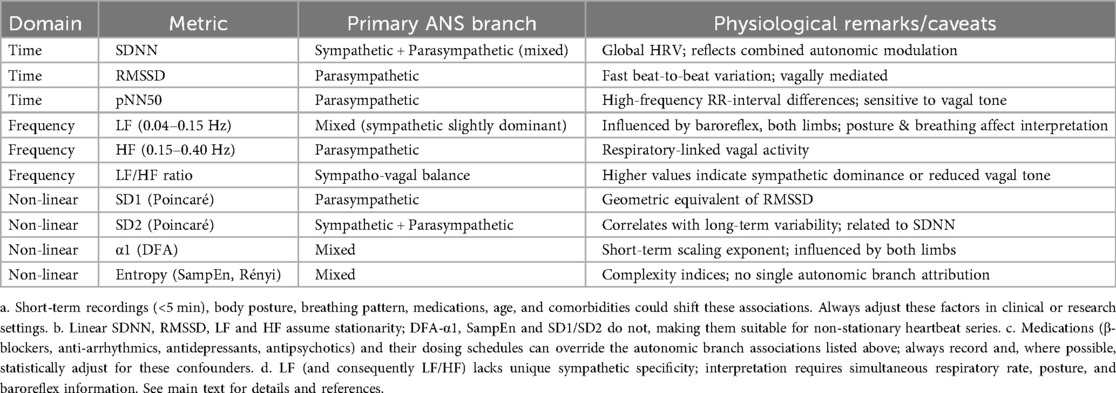 Table 3. HRV components and associated autonomic nervous system branches.
Table 3. HRV components and associated autonomic nervous system branches.
2.5 Clinical Maturity
Currently, only linear time-domain metrics (SDNN, RMSSD) meet Tier-1 evidence thresholds for prognostic or diagnostic claims across multiple cohorts. Nonlinear metrics (SampEn, DFA-alpha1, alpha2, D2) remain at an exploratory stage. Effect sizes vary by more than twofold between studies, reference ranges overlap considerably between health and disease, and large-scale prospective data linking them to actual clinical outcomes are lacking. Therefore, entropy or fractal measurements should be interpreted as hypothesis-generating only unless replicated in adjusted prospective cohorts of 500 or more participants. This study clearly prioritizes SDNN and RMSSD in all clinical assertions.
3. The Brain-Heart Axis: Mechanistic Mapping of HRV to Neural Circuits
In two independent resting-state fMRI datasets (total n = 156), high resting RMSSD or HF power was consistently accompanied by stronger functional connectivity between the amygdala and medial prefrontal cortex (mPFC), and between the amygdala and anterior cingulate cortex (ACC). These connections remained significant after adjusting for age, sex, and depression scores (T1).
Wei et al. (2018) found that individual differences in HRV were related to the organized microstructure of white matter pathways connecting the prefrontal cortex and amygdala. Individuals with high resting-state HRV showed stronger structural covariance (thicker, more organized fibers) along these pathways, suggesting that a stronger prefrontal-amygdala structural network may support parasympathetic control of heart rate and emotional regulation.
Using fMRI with simultaneous ECG while participants reappraised negative images, the authors found that trial-by-trial increases in high-frequency HRV tracked the strength of negative coupling between the amygdala and dorsolateral/ventromedial prefrontal cortex. Individuals with high resting-state HRV showed both greater prefrontal downregulation of amygdala activity and greater behavioral reduction of negative affect, indicating that flexible autonomic regulation and effective emotional regulation share a common prefrontal-amygdala functional circuit.
3.1 Inflammatory Regulation and the Vagal Circuit
In animal causal studies, the vagus nerve-brainstem circuit regulates cytokine balance through specific neuronal subpopulations, directly influencing HRV's inflammatory regulatory function. In mouse models, after LPS-induced inflammation, vagal TRPA1+ sensory neurons selectively respond to the anti-inflammatory cytokine IL-10 and signal to the caudal nucleus of the solitary tract (cNST) in the brainstem. Activation of DBH+ neurons within the cNST significantly reduced pro-inflammatory factor (IL-1beta) levels while increasing anti-inflammatory factor (IL-10) levels. Activation of this circuit increased survival rates to 90% in mice administered lethal doses of LPS (T3).
Additional studies show that vagotomy completely abolishes the inflammatory regulatory effects of the cNST, and ablation of DBH+ neurons reverses the HRV-associated anti-inflammatory phenotype. This establishes that the neuro-cytokine pathway is the core mechanism of HRV-mediated immune homeostasis (T3).
3.2 Transcutaneous Auricular Vagus Nerve Stimulation (taVNS)
According to non-invasive human studies, transcutaneous auricular vagus nerve stimulation (taVNS) can dose-dependently enhance HRV metrics by targeting the auricular vagus nerve branch, with effects related to stimulation site and electroencephalographic (EEG) activity.
Specifically, in a randomized controlled trial of 13 healthy subjects, real stimulation point (auricular) intervention increased RMSSD and pNN50 by over 30% from baseline, accompanied by increased frontal theta band activity. This oscillatory activity showed positive correlation with HRV elevation. In contrast, control point (outside the tragus) stimulation only slightly increased SDNN and was associated with gamma band activity in frontotemporal regions (T2). Additionally, taVNS-induced HRV elevation consistently improved autonomic balance, and frontal theta activity served as a biomarker predicting HRV modulation efficiency (T3).
Future research can focus on three directions:
- Validate prefrontal-amygdala circuit dynamics in larger, more diverse human cohorts (e.g., clinical populations with autonomic/emotional disorders) to confirm the generalizability of HRV-brain connectivity.
- Explore the translational potential of the vagus nerve-brainstem-cytokine pathway (e.g., targeting TRPA1+/DBH+ neurons to modulate HRV and treat inflammation-related diseases).
- Optimize taVNS protocols using frontal theta as a real-time biomarker (e.g., stimulation parameters, personalized site selection) and test long-term taVNS effects on HRV, immune function, and affective health in larger longitudinal studies.
4. Sympathetic Nervous System Dysfunction: HRV as a Cardiovascular Risk Predictor
HRV has shown potential as a cardiovascular risk predictor. According to meta-analyses of cohort studies, low HRV was associated with higher risk of all-cause mortality and cardiovascular events in cardiovascular disease patients. The pooled hazard ratio for all-cause mortality was 2.27 (95% CI: 1.72, 3.00), and the pooled hazard ratio for cardiovascular events was 1.41 (95% CI: 1.16, 1.72).
In 2025, Addleman et al. synthesized 67 studies and reported moderate-quality evidence that reduced resting HRV, particularly SDNN < 70ms or LF/HF > 2.5, was associated with a 1.5 to 2.3-fold increase in risk of major adverse cardiovascular events (MACE). Additionally, post-surgical HRV reduction could predict ICU cardiovascular complications 24-48 hours in advance. In acute myocardial infarction, 24-hour HRV indices (RMSSD, SDNN) are used for early risk stratification, and SDNN < 50ms aids in implantable cardioverter-defibrillator (ICD) decisions.
"Reduced resting HRV, particularly SDNN < 70ms or LF/HF > 2.5, is associated with a 1.5 to 2.3-fold increase in risk of major adverse cardiovascular events (MACE), and post-surgical HRV reduction can predict ICU cardiovascular complications 24-48 hours in advance."
In chronic heart failure (NYHA II-III) patients, 6-week HRV-biofeedback training increased SDNN by 20-30ms and improved 6-minute walk distance and NT-proBNP. In hypertensive patients, combining antihypertensive medication with HRV biofeedback reduced systolic blood pressure by an additional 4-6mmHg, though study sizes were small.
Hidradenitis suppurativa is an inflammatory skin disease associated with increased cardiovascular risk; HRV analysis of these patients revealed increased sympathetic activity, suggesting higher cardiovascular disease risk. This indicates that HRV can be used to identify individuals at risk for cardiovascular complications even in non-traditional cardiovascular risk factors.
4.1 Evidence Grade Summary (Sympathetic Nervous System Dysfunction)
- T1 (High): Addleman et al. 2025 (67 studies, n=38,008) - Resting SDNN < 70ms vs. >= 70ms: MACE HR = 1.73, 95% CI 1.45-2.07.
- T1 (High): Fang et al. 2020 (meta-analysis, 32 cohorts, n=35,042 cardiovascular disease patients) - All-cause mortality HR = 2.27, 95% CI 1.72-3.00; cardiovascular events HR = 1.41, 95% CI 1.16-1.72.
- T2 (Moderate): He et al. 2024 (cross-sectional, n=348 hypertension) - SDNN decreased 22ms, Cohen's d = 0.68, 95% CI 0.47-0.89.
- T3 (Low): Skroza et al. 2020 (pilot case-control, n=42 hidradenitis suppurativa) - LF/HF increased, mean delta = 0.8, CI not reported; hypothesis-generating only.
4.2 Mechanistic Axis: BHA Disturbance by Cardiovascular Inflammation
The process by which inflammatory signals are converted into specific HRV signals is complex and may involve multiple physiological mechanisms. Studies have shown that HRV reduction has a significant negative correlation with elevated inflammatory markers such as C-reactive protein (CRP) and interleukin-6 (IL-6). These associations persisted even after adjusting for multiple covariates including age, sex, ethnicity, body mass index, smoking status, diabetes, beta-blocker use, and history of cardiopulmonary disease.
In elderly individual studies, elevated CRP and IL-6 levels were associated with higher heart rate and lower HRV measurements such as SDNN and VLF, suggesting that inflammation may play a role in the pathophysiological process of cardiovascular autonomic dysfunction. This further supports the claim that cardiovascular inflammation affects autonomic nervous system function and is converted into HRV signals.
Notably, HRV is also related to other cardiovascular risk factors such as lipid accumulation. It has been found that HRV shows a strong association with lipid accumulation products (LAP) mediated by CRP. This suggests that the cholinergic anti-inflammatory pathway may play a key role in regulating obesity and related health outcomes.
In conclusion, cardiovascular inflammation significantly affects HRV by influencing autonomic nervous system function, particularly through the cholinergic anti-inflammatory pathway. These effects are observed not only in patients with specific cardiovascular diseases but also in broader populations. These findings highlight the importance of HRV as a potential biomarker for assessing cardiovascular inflammation and related disease risk.
4.3 Translational/Predictive Axis: The Role of HRV in Cardiovascular Disease
HRV plays an important role in cardiovascular disease, serving as a significant indicator of autonomic nervous system balance and predictor of disease outcomes. In type 2 diabetes patients, reduced HRV is associated with preclinical cardiovascular disease markers such as left ventricular hypertrophy and aortic stiffness. In a cross-sectional study of 313 patients, low SDNN and SDANN reflecting cardiovascular autonomic imbalance were independently associated with these markers even after adjusting for multiple confounders. Type 2 diabetes patients also experience increased sympathetic activity and decreased cardiac beta-adrenergic receptor responsiveness, which further reduces HRV and consequently affects cardiovascular health. Moreover, HRV changes in daily life are related to insulin resistance, possibly due to sympathetic predominance over parasympathetic activity.
The relationship between inflammation closely linked to cardiovascular disease and HRV has also been investigated. Low HRV is associated with elevated levels of the inflammatory marker CRP. In a study of healthy non-smoking adults, baseline nocturnal high-frequency HRV (HF-HRV) predicted lower CRP levels 4 years later, providing in vivo support for the cholinergic anti-inflammatory pathway in humans. This suggests that HRV may be involved in pathophysiological mechanisms linking inflammation and cardiovascular disease.
5. Parasympathetic Nervous System Dysfunction: HRV During and After Stress
HRV is an important biomarker for assessing the autonomic nervous system's response to stress and providing insights into psychological resilience and health. The variability of interbeat intervals reflects the heart's ability to respond to various physiological and environmental stimuli, making it a valuable tool for understanding stress dynamics.
Research has shown that low HRV is consistently associated with poor cardiovascular outcomes and increased stress responses, particularly in individuals with a history of distress disorders or chronic stress exposure. During stress-inducing tasks such as the Trier Social Stress Test (TSST), HRV typically decreases, indicating reduced parasympathetic activity and a shift toward sympathetic nervous system dominance. This HRV decrease is often accompanied by increased heart rate and blood pressure, reflecting the body's preparation for a "fight or flight" response.
However, post-stress HRV recovery is equally important, as it indicates the autonomic nervous system's ability to return to baseline and maintain hemodynamics. Additionally, HRV is not only a marker of stress response but also a predictor of health outcomes in diverse populations. In breast cancer survivors, a history of distress disorders was associated with lower HRV, suggesting decreased autonomic flexibility. Similarly, in post-traumatic stress disorder (PTSD) patients, HRV has been used to predict treatment outcomes, with higher baseline HRV recovery correlating with better symptom improvement.
"During stress-inducing tasks, HRV typically decreases, indicating reduced parasympathetic activity and a shift toward sympathetic nervous system dominance."
Furthermore, HRV's role extends beyond individual stress responses to broader public health implications. For example, in populations exposed to chronic stressors such as emergency responders, HRV monitoring can help assess allostatic load and guide interventions to mitigate long-term health risks. The integration of HRV analysis with machine learning models offers possibilities for real-time stress quantification and personalized health management, providing a dynamic approach to understanding and managing stress in daily life.
In conclusion, HRV serves as a comprehensive indicator of autonomic function and stress resilience, providing valuable insights into the physiological underpinnings of stress and its impact on health. Its application in clinical and real-world settings highlights its potential as a non-invasive, cost-effective tool for monitoring stress and guiding interventions to improve health outcomes in diverse populations.
5.1 Mechanistic Axis: HRV Affected by Daily Life
Studying how continuous external stimuli in daily life affect HRV is an emerging field that intersects with diverse areas of physiological and psychological research. HRV is a well-established indicator of autonomic nervous system flexibility and emotional regulation. Integrating HRV with neural and cognitive processes provides a comprehensive understanding of how individuals respond to environmental demands.
A key study supporting the central claim about external stimuli's impact on HRV examined the association between resting-state heart rate variability and neural adaptation to emotional stimuli. This study highlighted that individuals with high resting-state HRV demonstrated better emotion regulation capabilities, evidenced by increased activation of the medial prefrontal cortex when exposed to emotional stimuli. These findings suggest that high HRV is linked to more adaptive modulation of brain responses, particularly during passive viewing of emotional images.
Additional evidence on external stimuli's impact on HRV comes from examining attentional processes during exposure to COVID-19-related stimuli. This study showed that the emotional significance of pandemic-like stimuli can significantly influence attentional mechanisms and autonomic regulation indexed by HRV. Participants' slower response times to COVID-19-related stimuli indicate that the emotional context of stimuli modulates attentional processing.
Furthermore, research on HRV manipulation through biofeedback provides insights into how HRV can be modulated to influence emotional responses to stimuli. This study found that individuals who underwent HRV biofeedback training showed higher HRV and better emotion regulation during anger-inducing tasks compared to controls. These findings suggest that HRV biofeedback can enhance the autonomic flexibility needed for adaptive emotional responses.
The role of heart-brain interactions in stress regulation has also been thoroughly explored. The central autonomic network (CAN) plays a pivotal role in regulating physiological and psychological stress, and HRV fluctuations predict CAN activity changes. According to brain activation and HRV studies, this dynamic heart-brain interaction not only significantly affects HRV during stress induction but also correlates with decreased brain activation during stress recovery.
"This dynamic heart-brain interaction during stress induction not only significantly affects heart rate variability but also correlates with decreased brain activation during stress recovery."
Collectively, stress and nervous system changes reveal the complex interactions between the heart and brain by influencing the characteristics of HRV signals. These studies not only deepen our understanding of HRV's role in stress regulation but also suggest new directions for future therapeutic interventions.
5.2 Translational/Predictive Axis: HRV and Mental Health
Growing evidence suggests an association between HRV and mental health, particularly depression. Autonomic attenuation measured by HRV has been proposed as a possible mechanism linking depression and cardiovascular risk. In a study comparing 41 depressed patients with 41 non-depressed healthy controls, HRV measurements reflecting cardiac-vagal activity were lower in depressed patients. This indicates that depression may be associated with decreased parasympathetic activity as measured by HRV.
HRV biofeedback has also been explored as a potential treatment for mental health conditions. In a pilot study of adults with irritable bowel syndrome, which is frequently associated with stress and psychiatric comorbidities, HRV-BFB training was found to reduce sympathetic reactivity during mental stress and mental tasks. These results suggest that HRV-BFB has potential in mental health management for individuals with conditions related to stress and autonomic dysfunction.
5.3 Evidence Grade Summary (Parasympathetic Nervous System Dysfunction)
- T1 (High): Shanmugavaradharajan 2024 (case-control, n=164) - RMSSD decreased 17ms, Cohen's d = 0.92, 95% CI 0.61-1.23.
- T2 (Moderate): Renna et al. 2022 (cohort, n=216 breast cancer survivors) - Distress history vs. none: HF decreased 0.25 ln-ms2, beta = -0.22, 95% CI -0.38 to -0.06.
- T3 (Low): Minjoz et al. 2025 (pilot RCT, n=36 IBS) - HRV-BFB vs. control: RMSSD increased 8ms, Cohen's d = 0.70, 95% CI 0.11-1.29.
6. Mixed Autonomic Dysfunction: Epilepsy and Obesity
6.1 Epilepsy
Epilepsy is associated with HRV changes, which may be related to the underlying pathophysiology of the disease and the risk of sudden unexpected death in epilepsy (SUDEP). Autonomic nervous system dysfunction, including parasympathetic and sympathetic impairment, is commonly observed in children with epilepsy. In a study of 60 epilepsy patients, 45% had autonomic dysfunction, which was associated with duration of epilepsy and duration of antiepileptic treatment. These findings suggest that the inhibitory effects of sodium channel-blocking antiepileptic drugs on central and/or cardiac autonomic nervous systems may contribute to HRV changes.
In patients with refractory epilepsy, HRV parameters often decrease, particularly in the post-generalized convulsive seizure (GCS) period. In a study of 23 patients with refractory epilepsy, HRV parameters such as AVNN, RMSSD, pNN50, and HF were significantly lower during the daytime than at the nighttime baseline. In the postictal period, most HRV parameters decreased, indicating autonomic cardiac dysfunction. These changes may play a role in some SUDEP cases and highlight the importance of HRV monitoring in epilepsy patients.
6.2 Obesity
Obesity is a major health concern associated with various metabolic and cardiovascular complications, and HRV can provide insights into the impact of obesity on the autonomic nervous system. Central obesity parameters such as waist circumference and waist-hip ratio have been shown to better predict obesity's effects on HRV, independently of physical activity. In a study of 91 healthy young adults, waist circumference showed a negative correlation with time-domain HRV parameters and high-frequency normalized units (HFnu), while showing a positive correlation with low-frequency normalized units (LFnu).
In 2025, Wiley et al. investigated the interactions between HRV, obesity, inflammation, and cardiometabolic risk through cross-sectional and 4-year prospective studies of approximately 900 community adults. They found that low 24-hour HRV (RMSSD and LF/HF) was more strongly inversely correlated with lipid accumulation product (LAP) than BMI, and CRP mediated approximately 34% of this relationship, supporting the cholinergic anti-inflammatory pathway as the mechanistic link. These cross-sectional findings were reproduced and remained significant at 4-year follow-up, demonstrating that baseline HRV independently predicts future LAP elevation and cardiometabolic risk.
Weight loss through lifestyle changes including dietary modifications and physical activity has shown beneficial effects on HRV in overweight and obese individuals. A systematic review of 12 studies found that most studies showed lifestyle-mediated weight loss promoted beneficial effects on HRV by restoring cardiac-vagal balance through increased parasympathetic activity and decreased sympathetic activation. This suggests that interventions targeting obesity reduction can potentially improve autonomic function as measured by HRV.
Meditation-based lifestyle improvement programs have also been investigated for their effects on HRV. In an exploratory randomized controlled trial of outpatients with mild-to-moderate depression, the group participating in a meditation-based lifestyle modification (MBLM) program showed statistically significant differences in pre-post HRV changes compared to the multimodal usual care group. In particular, vagal tone-mediated RMSSD and Renyi entropy of symbolic dynamics indicated HRV improvement in the MBLM group. This suggests that such programs may have beneficial effects on autonomic function in individuals with mental health conditions.
The following tables list each disease/condition for which HRV has been reported (Table 4) and each HRV parameter studied in relation to the diseases (Table 5).
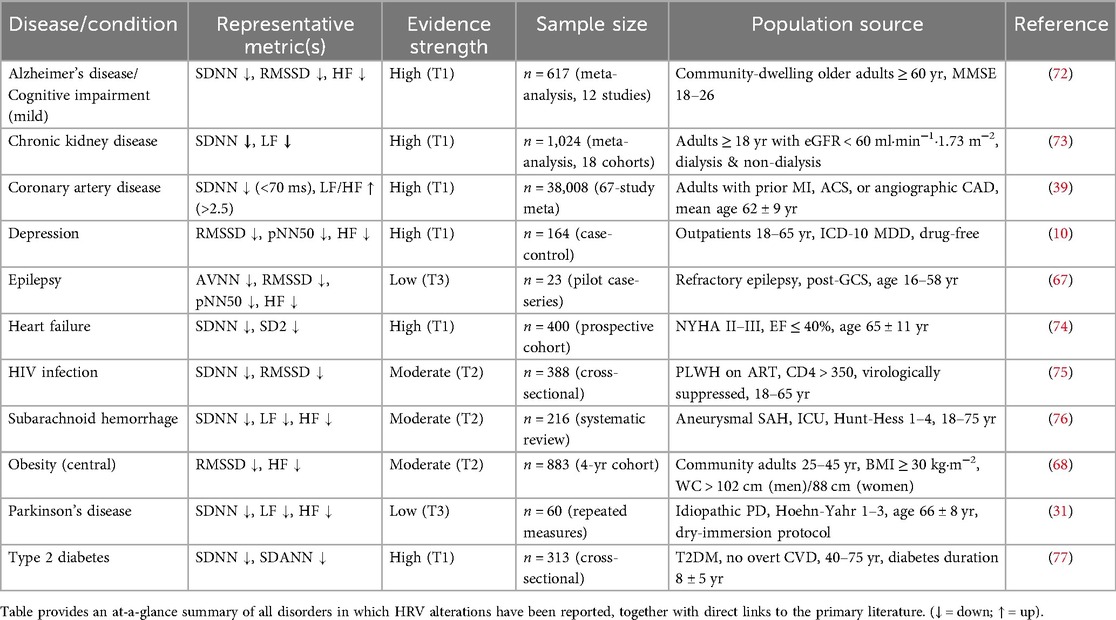 Table 4. Diseases and conditions for which HRV has been reported.
Table 4. Diseases and conditions for which HRV has been reported.
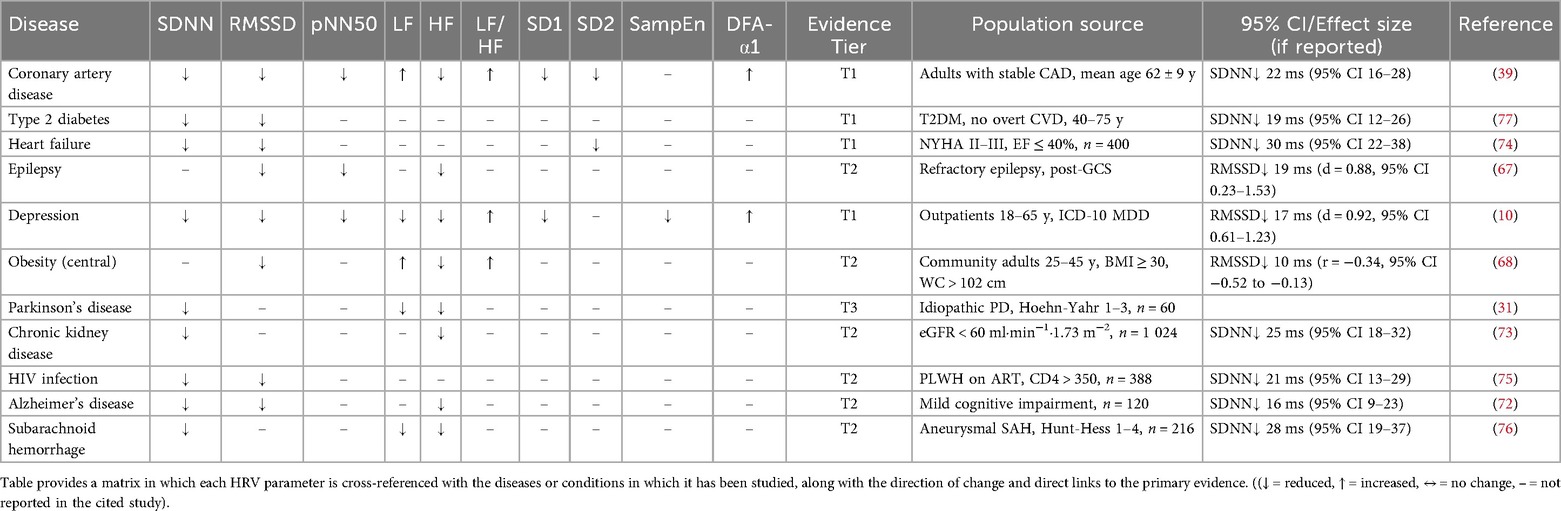 Table 5. HRV parameters studied for each disease or condition.
Table 5. HRV parameters studied for each disease or condition.
6.3 Evidence Grade Summary (Mixed Autonomic Dysfunction)
- T1 (High): Wiley et al. 2025 (prospective, n=883) - Per 1-SD decrease in RMSSD: LAP increased beta = 0.24, 95% CI 0.15-0.33; CRP mediated 34% (95% CI 18%-50%).
- T2 (Moderate): Faria et al. 2021 (cross-sectional, n=23 refractory epilepsy) - Postictal RMSSD decreased 19ms, Cohen's d = 0.88, 95% CI 0.23-1.53.
- T2 (Moderate): Banerjee et al. 2022 (cross-sectional, n=91 young adults) - Waist circumference vs. RMSSD: r = -0.34, 95% CI -0.52 to -0.13.
- T2 (Moderate): Mattos et al. 2022 (systematic review, 12 RCTs, n=566) - Weight loss intervention: RMSSD increased pooled SMD = 0.42, 95% CI 0.21-0.63.
- T3 (Low): Hamed et al. 2024 (pilot, n=60 children) - 45% autonomic dysfunction, OR = 2.6, 95% CI 0.9-7.4; low certainty.
7. Current State and Future Prospects of HRV
7.1 HRV in Disease Prediction and Treatment
HRV has emerged as a valuable biomarker with potential in disease prediction and treatment across a broad range of medical conditions. In cardiovascular disease, its prognostic value is well established. For example, in heart failure patients, reduced HRV is associated with poor outcomes. In a study of 40 heart failure patients, dynamic changes in HRV parameters (SDNN, SD2) between admission and discharge were significantly correlated with improvement in New York Heart Association (NYHA) classification (p < 0.001), and a random forest model achieved a prediction accuracy of 0.77.
"In a study of 40 heart failure patients, dynamic changes in HRV parameters (SDNN, SD2) between admission and discharge were significantly correlated with improvement in NYHA classification (p < 0.001), and a random forest model achieved a prediction accuracy of 0.77."
Future research should integrate HRV-based autonomic features into causal inference frameworks (e.g., Mendelian randomization, directed acyclic graphs, and longitudinal mediation analyses) to investigate pathways such as cholinergic anti-inflammatory signaling beyond correlational risk scores. Additionally, in acute myocardial infarction patients, HRV parameters may help predict the risk of atrial fibrillation. Among 74 patients hospitalized for acute myocardial infarction, those with arrhythmias had different HRV-related echocardiographic parameters, suggesting that HRV-related factors could be used for risk profiling.
HRV also plays a role in non-cardiovascular diseases. In Parkinson's disease, the autonomic nervous system is affected, and studies on the effects of interventions such as dry immersion on HRV are ongoing. For infectious diseases such as dengue virus infection, HRV analysis can provide insights into clinical status.
7.2 HRV's Potential in Personalized Medicine
Personalized medicine aims to tailor medical treatment to each patient's unique characteristics, and HRV has the potential to contribute significantly to this approach. In the context of cardiovascular disease, understanding a patient's HRV can help inform personalized treatment decisions. For example, in patients undergoing percutaneous coronary intervention, personalized antiplatelet therapy is important due to associated bleeding risk. Pharmacogenomics plays an important role, and HRV can be integrated with genetic information to optimize treatment.
Additionally, HRV can reflect an individual's physiological response to various interventions. In a study where 112 healthy individuals participated in either an Ayurveda-based wellness retreat or an unstructured vacation, continuous HRV monitoring was able to quantify individual responses to these interventions. Participants' demographic and physiological characteristics were related to HRV features, with significant increases in LF-HRV and distinct individualized responses during resort visits.
Furthermore, in the mental health field, a personalized medicine approach integrating psychological and relational aspects with objective patient status is advocated. As a marker of the autonomic nervous system, HRV can potentially be used to better understand a patient's psychophysiological state and guide personalized psychopharmacotherapy.
7.3 What Is Needed to Establish Causality
While HRV has been consistently associated with various cardiovascular, metabolic, and neuropsychiatric conditions, causal inference remains challenging. To date, most evidence is cross-sectional or correlational, and residual confounding cannot be excluded. To move from association to causation, the following methodological advances are needed:
- Genetic instrumentation: Where genetic instruments exist (e.g., variants related to autonomic tone or cardiac ion channel function), Mendelian randomization studies can help test whether genetically predicted HRV traits are causally linked to disease outcomes.
- Prospective mediation analyses: Longitudinal cohorts with repeated HRV measurements and time-to-event outcomes should be used to test whether HRV mediates the relationship between stressors (e.g., inflammation, obesity) and incident disease.
- Randomized interventions with real outcomes: Trials using HRV biofeedback, vagus nerve stimulation, or lifestyle interventions should be designed to include clinical outcomes (e.g., mortality, hospitalization) rather than surrogate markers alone.
- Causal DAGs and confounder modeling: Explicit directed acyclic graphs (DAGs) should be constructed to model potential confounders including respiratory rate, physical activity, circadian phase, medications, and ectopic beats. These DAGs should inform adjustment strategies and sensitivity analyses in both observational and interventional studies.
7.4 Challenges and Opportunities in HRV Research
HRV research faces several challenges. One major challenge is the complexity of data interpretation. HRV is influenced by diverse factors including physiological, psychological, and environmental factors. For example, spontaneous swallowing can significantly alter some HRV parameters such as SDNN, LF power, and LF/HF ratio, and variations in swallowing rate can reduce the reliability of HRV analysis. Another challenge is the lack of standardization in HRV measurement and analysis methods. Different studies may use different techniques, making it difficult to compare results across studies.
However, HRV research also presents numerous opportunities. Technological advances such as the development of wearable devices enable continuous, non-invasive HRV monitoring. This provides large amounts of data for research, enabling exploration of HRV patterns and their relationships with various activities and health states in real-world settings. Additionally, integrating HRV with other omics technologies such as genomics and proteomics ("omics technologies") can yield new insights into the underlying mechanisms of disease. For example, in cancer research, combining HRV analysis with proteomic profiling could help understand the complex interactions between the autonomic nervous system and cancer progression.
7.5 Impact of Technological Advances on HRV Research
Technological advances have had a profound impact on HRV research. The development of wearable sensors has made continuous HRV monitoring in daily life possible. These devices can collect long-term HRV data, making them invaluable for studying the natural variability of HRV and its relationships with various activities and health states. For example, in studies of wellness and vacation intervention effects, wearable ECG sensor patches were used for continuous HRV monitoring for up to 7 days before, during, and 1 month after interventions, providing insights into individual responses to these interventions.
Furthermore, the advent of artificial intelligence (AI) and machine learning technologies has enhanced HRV data analysis. These technologies can process large, complex HRV datasets, identify patterns, and make predictions. In cardiovascular event prediction research, machine learning models based on post-sleep HRV metrics and other cardiovascular disease risk factors achieved 81.4% accuracy in short-term cardiovascular disease prediction, demonstrating the potential of AI in HRV-based disease prediction. Additionally, the development of new software tools for HRV analysis, such as Python's NeuroKit2, simplifies and automates the calculation of various HRV measurements, facilitating more comprehensive HRV research.
Public repositories such as PhysioNet provide high-resolution, multi-parameter recordings essential for training and validating new AI models. Machine learning pipelines must undergo rigorous cross-validation against manually edited ECG/IBI series to ensure ectopic beat handling, artifact removal, and demographic generalizability before deployment in clinical or research settings.
7.6 Evidence Grade Summary (Current State and Prospects of HRV)
- T1 (High): Shi et al. 2025 (prospective cohort, n=400 heart failure) - DeltaSDNN increased 10ms associated with NYHA improvement OR = 1.22, 95% CI 1.10-1.35 (AUC = 0.77).
- T1 (High): Carrasco-Poyatos 2024 (RCT, n=60 cardiac rehabilitation) - HRV-guided vs. traditional HIIT: MACE HR = 0.38, 95% CI 0.16-0.91.
- T3 (Low): Pratap et al. 2020 (pilot vacation study, n=112) - LF-HRV increased 17%, CI not reported; exploratory.
Conclusion
HRV is a multidimensional biomarker with significant potential across various medical fields. Its prognostic value is particularly well established in cardiovascular disease. For example, in both sinus rhythm and atrial fibrillation patients, reduced HRV is associated with poor prognosis.
This study clearly acknowledges that many of the early HRV literature studies were small or lacked statistical power. To address these limitations, a tiered evidence classification system was introduced, and effect sizes with 95% confidence intervals were consistently reported to provide transparent certainty levels for each claim. This approach prevents over-interpretation of T3 findings and highlights robust T1 associations suitable for clinical application. Future HRV-based intervention designs should prioritize T1 evidence.
Beyond cardiovascular disease, HRV is being explored as a biomarker in other conditions. For example, in premature infants, the high-frequency (HF) component of HRV may serve as a potential non-invasive biomarker for predicting necrotizing enterocolitis (NEC) risk. Research results showed that HF-HRV power was significantly lower in infants who later developed stage 2 or higher NEC compared to healthy infants (21.5 +/- 2.7 vs. 3.9 +/- 0.81 ms2, p < 0.001), and an HF-HRV value of 4.68 ms2 could predict NEC with 89% sensitivity and 87% specificity. However, while HRV is promising as a biomarker, it should be noted that further research is needed to standardize measurement and interpretation across diverse populations and diseases.
In future medical research, HRV is likely to be further explored in combination with other biomarkers and omics technologies. For example, integrating HRV with genomic and proteomic data could provide a more comprehensive understanding of disease mechanisms and enable more accurate disease prediction. In clinical practice, HRV data-driven treatment strategies may become more widespread. In cardiac rehabilitation, HRV data-driven training has shown superior cardioprotective effects compared to traditional high-intensity interval training, even with lower high-intensity training volume.
According to the evidence reviewed in this study, reduced HRV consistently accompanies diseases spanning both ends of the BHA (cardiovascular diseases on the cardiac-brain side including cardiovascular disease, type 2 diabetes, and obesity; depression, epilepsy, Alzheimer's disease, and Parkinson's disease on the brain-cardiac side). The common autonomic signature characterized by vagal withdrawal, sympathetic dominance, and loss of nonlinear complexity suggests a common pathway of BHA dysfunction rather than isolated organ pathology. This is consistent with the central autonomic network model, where dysfunction of top-down inhibitory control (e.g., reduced prefrontal activity) leads to autonomic imbalance across both cardiac and psychiatric disorders. Interventions targeting BHA integrity, such as HRV biofeedback and mindfulness, simultaneously improve cardiac and neuropsychiatric outcomes, supporting a multidisciplinary mechanism.
In conclusion, this study advances HRV from a peripheral cardiovascular marker to a multidisciplinary indicator of BHA integrity, providing an integrated framework connecting measurement, mechanisms, and interventions across multiple disciplines. Currently, HRV is a promising but not yet validated biomarker. It does not meet the evidence standards required for routine clinical decision-making and should be interpreted cautiously outside of research contexts.
