This study analyzed longitudinal multi-omics data from 108 participants and revealed that the molecular changes occurring during human aging are largely nonlinear. In particular, it discovered a "wave" phenomenon in which aging-related molecular and functional changes are dramatically amplified at two major transition points around ages 40 and 60. These nonlinear changes are associated with increased risk of various diseases including cardiovascular disease, kidney disease, and type 2 diabetes, as well as declining immune function, providing important insights for the early diagnosis and prevention of aging-related diseases.
1. Aging Is Not Linear: Nonlinear Changes in Molecules and Microbiome
Human aging is a complex, multifactorial process that extends beyond simple physiological changes, closely linked to various diseases including cardiovascular disease, diabetes, neurodegenerative diseases, and cancer. Recent advances in high-throughput omics technologies that can systematically study changes at various molecular levels -- genes, proteins, metabolites, cytokines -- have been tremendously helpful in understanding the fundamental mechanisms of aging and discovering therapeutic targets. However, most studies have viewed molecular changes with aging from a linear perspective.
Interestingly, the incidence of aging-related diseases does not simply increase proportionally with age but shows nonlinear patterns that accelerate at specific time points. For example, in the United States, the prevalence of cardiovascular disease is approximately 40% among those aged 40-59, but surges to approximately 75% among those aged 60-79 and approximately 86% among those aged 80 and above. Neurodegenerative diseases such as Parkinson's and Alzheimer's also show distinct transition points around ages 40 and 65, with prevalence tending to increase. Research has also shown that brain aging accelerates in middle and old age. These nonlinear increases in disease prevalence strongly suggest that the human aging process is not simply linear.
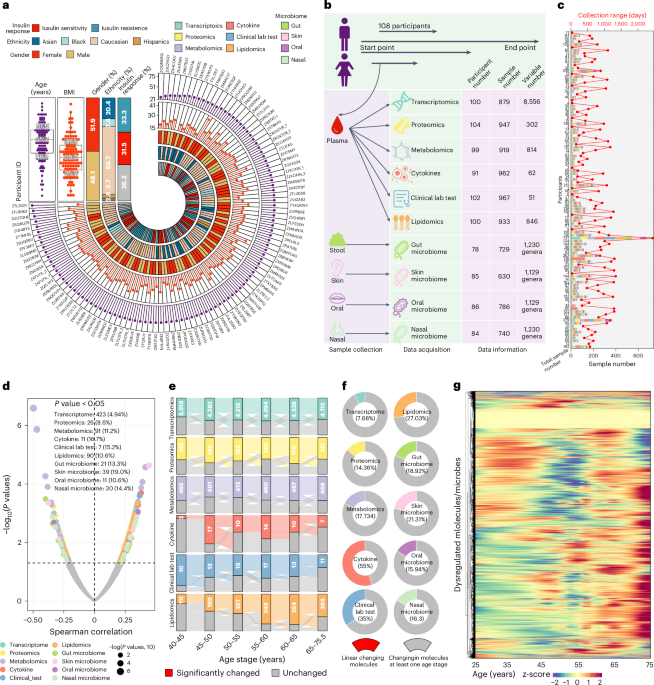
In this study, longitudinal multi-omics profiling was performed on 108 participants between the ages of 25 and 75. These participants were followed for an average of 1.7 years (up to 6.8 years), with various omics data (transcriptome, proteome, metabolome, cytokines, clinical laboratory tests, lipidome, microbiome, etc.) collected from blood, stool, skin, oral, and nasal samples. A total of 135,239 biological features were analyzed from 5,405 biological samples, yielding over 246.5 billion data points. Such a vast longitudinal dataset is highly effective at capturing the complex molecular and functional changes that occur during the aging process.
Remarkably, the analysis found that only a very small fraction (6.6%) of all molecules and microbes showed linear changes. In contrast, the vast majority -- 81% of molecules and microbes -- exhibited nonlinear change patterns during aging. This suggests that existing linear models are insufficient to fully understand the complexity of aging, underscoring the widespread nonlinear nature of aging-related molecular changes.
"The majority of molecules and microbes undergo nonlinear changes during the human aging process."
2. Discovering Nonlinear Multi-Omics Change Patterns Through Clustering
The research team evaluated how well the multi-omics data collected from the longitudinal cohort reflected the aging process. Analysis showed that many omics datasets had significant correlations with participants' ages. In particular, metabolome, cytokine, and oral microbiome data showed the strongest associations with age, indicating that these data could serve as powerful indicators of the aging process.
To compensate for the fact that omics data do not align precisely, the team smoothed the data using previously published methods. Then, to reveal specific patterns of molecules changing during aging, they used unsupervised fuzzy c-means clustering to group molecules with similar trajectories. This allowed them to identify 11 molecular trajectory clusters that change over the course of aging.
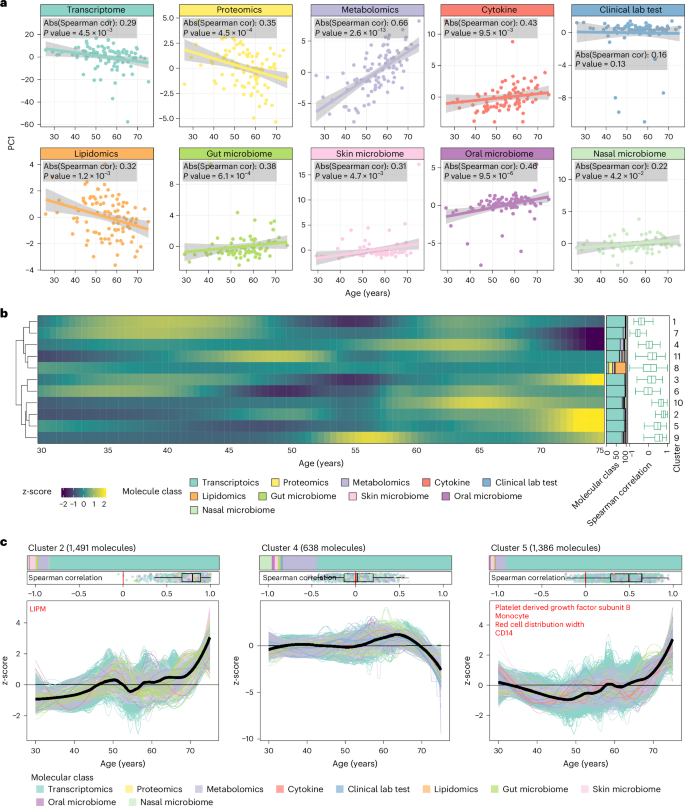
Among the 11 clusters, clusters 2, 4, and 5 showed clear and interpretable patterns across the lifespan.
- Cluster 4: Maintained a relatively stable pattern until around age 60, then showed a sharp decline afterward.
- Clusters 2 and 5: Showed variability before age 60, then exhibited sharp increases or upward inflection points around ages 55-60.
These cluster patterns once again strongly suggest that aging is not a linear process.
An interesting point is that female menopause typically occurs between ages 45 and 55, which is very close to the major transition points observed in these three clusters. However, when clustering analysis was performed separately by sex, similar clusters appeared in both male and female datasets. This means that the transition point around age 55 is not limited to female menopause but is a common aging phenomenon in both sexes.
Additionally, to check whether transcriptome data might bias the results, two additional clustering analyses were performed -- one using only transcriptome data and one excluding it. Remarkably, both analyses showed nearly identical cluster configurations to those found using the full omics dataset, confirming that the identified clusters are consistent across various omics platforms and are not driven by transcriptome data.
3. Nonlinear Changes in Function and Disease Risk with Aging
To further investigate the biological functions associated with the nonlinearly changing molecules in the three major clusters (2, 4, and 5), the research team performed individual functional analyses on the transcriptome, proteome, and metabolome datasets. This allowed them to more precisely define the change patterns of previously reported functional modules and to discover new, previously unreported potential functional modules.
3.1. Transcriptome Functional Changes
- Cluster 2:
- Increased GTPase activity: Closely related to apoptosis (programmed cell death), consistent with previous findings that this activity increases with aging.
- Increased histone modification: Also known to increase with aging.
- Cluster 4:
- Decreased oxidative stress-related modules: Including antioxidant activity, oxygen transport activity, oxygen binding, and peroxidase activity, linked to increased oxidative stress and ROS (reactive oxygen species) that positively influence aging-related inflammation.
- Cluster 5:
- mRNA stability-related modules: Related to mRNA destabilization, mRNA processing, and positive regulation of mRNA catabolic processes, consistent with previous research linking mRNA turnover to aging.
- Increased autophagy: Known to increase as aging progresses.
3.2. Functional Modules Related to Increased Disease Risk
Modules suggesting nonlinear increases in specific disease risks with aging were also found across several clusters.
- Cluster 2 (sharp increase after age 60):
- Phenylalanine metabolism pathway: Research shows that gradually increasing plasma phenylalanine levels are associated with cardiac dysfunction and act as a factor deviating from the cardiac aging trajectory.
- C-X-C motif chemokine 5 (CXCL5): Detected in proteome data, known to be present at high concentrations in atherosclerosis.
- Blood urea nitrogen (BUN): A clinical test indicator of kidney function, suggesting nonlinear decline in kidney function with aging.
- Serum/plasma glucose: An indicator of type 2 diabetes (T2D), consistent with the nonlinear increase in T2D risk after age 60.
"After age 60, the risks of cardiovascular disease, kidney disease, and type 2 diabetes surge nonlinearly."
- Cluster 4 (sharp decrease after age 60):
- Decreased unsaturated fatty acid biosynthesis pathway: Since unsaturated fatty acids help reduce cardiovascular disease risk and maintain brain function, a decline in this pathway implies increased cardiovascular disease risk.
- Decreased alpha-linolenic acid and linolenic acid metabolism pathways: These are fatty acids that help reduce aging-related diseases such as cardiovascular disease.
- Caffeine metabolism pathway: Suggests that caffeine metabolism capacity decreases with aging.
- Cluster 5:
- Mean corpuscular hemoglobin and red cell distribution width: These clinical test results are consistent with transcriptome data suggesting nonlinear decline in oxygen transport capacity during aging.
Additionally, cluster 11 showed a nonlinear change in which DNA repair capacity improved until age 50, then sharply declined between ages 50 and 56 before stabilizing. This demonstrates that DNA repair-related pathways important for genomic stability, gene expression, and metabolic balance change differently with aging.
Overall, these functional analyses provide valuable insights into how nonlinear changes in molecular profiles during aging relate to biological function and disease risk. In particular, they revealed that individuals over age 60 become more vulnerable to cardiovascular disease, kidney problems, and type 2 diabetes, with important implications for the diagnosis and prevention of these diseases.
4. Discovery of Wave Phenomena in Aging-Related Molecules
While the trajectory clustering method described above is effective at identifying nonlinearly changing molecules with clear, consistent patterns during aging, it may have limitations in capturing abrupt changes that occur only at specific age ranges. Therefore, the research team used a modified DE-SWAN algorithm to comprehensively understand changes in multi-omics profiles during aging. This algorithm compares 20-year windows in overlapping 10-year increments to identify molecules and microbes that become dysregulated with age.
Through this method, they discovered that thousands of molecules show specific change patterns during aging, forming distinct "waves." In particular, two prominent crests were observed around ages 45 and 65. This is consistent with results from previous studies conducted using only proteome data, and the second crest (around age 60) also aligns with the transition point found in the trajectory clustering results described above.
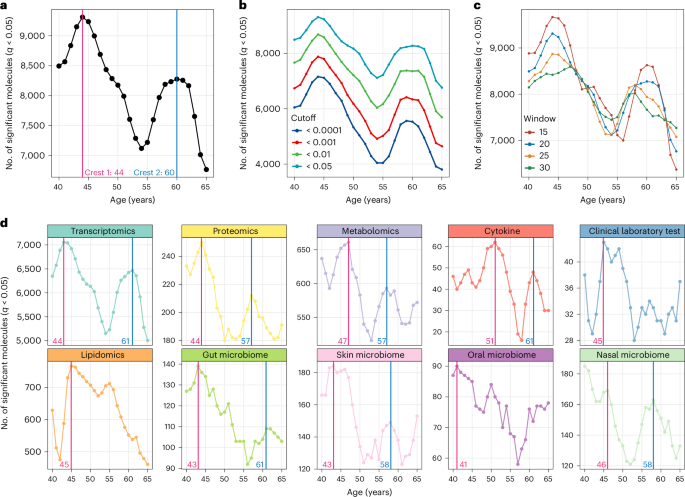
To validate the significance of these two crests, the research team performed analyses using various statistical parameters, and the same waves appeared consistently. Furthermore, when age data was randomly shuffled, these crests disappeared, supporting that these waves represent genuine characteristics of aging-related molecular changes.
Even more remarkably, these waves appeared across most omics data types, including transcriptome, proteome, metabolome, cytokines, gut microbiome, skin microbiome, and nasal microbiome. This supports the hypothesis that aging-related changes are not confined to specific omics layers but involve coordinated, systemic changes across multiple molecular components. The discovery of these consistent crests underscores the robustness and reliability of molecular milestones occurring during the aging process.
4.1. Functional Changes and Disease Risk at the Two Crests
Analysis of the roles and functions of molecules that become dysregulated at the two crests (the first crest around age 45, the second around age 65) identified several functional modules indicating nonlinear increases in aging-related disease risk.
-
Functional modules found at both crests:
- Cardiovascular disease (CVD)-related: Dysregulation of platelet degranulation, complement cascade, complement and coagulation cascades, protein activation cascades, and protease binding can affect the cardiovascular system in various ways and contribute to CVD. Dysregulation of blood coagulation and fibrinolysis processes, essential functions for maintaining blood fluidity, can also lead to thrombotic and cardiovascular events.
- Skin and muscle stability-related: Dysregulation of extracellular matrix (ECM) structural components, glycosaminoglycan (GAG) binding, and phosphatidylinositol binding suggests that skin and muscle aging accelerates markedly during these periods. Modules affecting muscle structure and function, including actin binding, actin filament organization, and actin cytoskeleton regulation, were also identified.
- Caffeine metabolism: Suggests that caffeine metabolism capacity changes notably around both ages 40 and 60.
-
Functional modules predominantly found at the first crest (around age 45):
- Lipid and alcohol metabolism: Lipid metabolism-related modules including plasma lipoprotein remodeling, chylomicron assembly, and ATP-binding cassette (ABC) transporters, as well as alcohol metabolism-related modules (alcohol binding) were identified. This suggests that lipid and alcohol metabolism efficiency declines around age 40.
"Around age 40, lipid and alcohol metabolism efficiency begins to decline."
- Lipid and alcohol metabolism: Lipid metabolism-related modules including plasma lipoprotein remodeling, chylomicron assembly, and ATP-binding cassette (ABC) transporters, as well as alcohol metabolism-related modules (alcohol binding) were identified. This suggests that lipid and alcohol metabolism efficiency declines around age 40.
-
Functional modules predominantly found at the second crest (around age 65):
- Immune dysfunction: Modules related to immune decline (immunosenescence) appeared, including acute phase response, antimicrobial humoral response, zymogen activation, complement binding, monocyte differentiation, viral processes, and regulation of hematopoiesis. This suggests a sharp decline in immune function after age 60.
- Kidney function: Kidney function-related modules such as glomerular filtration were identified, consistent with previous research indicating kidney function decline around age 60.
- Carbohydrate metabolism: Carbohydrate metabolism-related modules such as carbohydrate binding were also found, suggesting that the impact of carbohydrates on aging manifests during this period.
"After age 60, immune function declines sharply, and abnormalities in kidney function and carbohydrate metabolism also emerge."
These results demonstrate that the trajectory clustering approach and the DE-SWAN algorithm complement each other in providing a comprehensive understanding of nonlinear molecular and functional changes during human aging.
5. Conclusions and Future Research Directions
This study clearly demonstrated through analysis of longitudinal multi-omics data from 108 participants that molecular changes during human aging are largely not linear. In particular, it identified two major "crest"-shaped transition points occurring around ages 40 and 60, meaning that aging-related molecular and functional changes are significantly amplified during these periods.
- Key findings:
- 81% of aging-related molecules showed nonlinear changes, while only 6.6% showed linear changes.
- Metabolome, cytokine, and oral microbiome data best reflected age.
- Molecular trajectory clustering confirmed three distinct patterns showing abrupt changes around age 60. These transition points appeared commonly in both sexes.
- At the major transition points (around ages 40 and 60), functional modules associated with nonlinear increases in cardiovascular disease, kidney disease, and type 2 diabetes risk, as well as immune decline and various other disease and functional abnormalities, were identified. This vulnerability was particularly pronounced in those over 60.
- Skin and muscle aging also tended to accelerate around ages 40 and 60.
These findings can provide important insights for a more nuanced understanding of the aging process and for developing early diagnosis and prevention strategies for age-related diseases. Beyond simply growing older, by focusing on the dramatic molecular changes occurring at specific ages, this research holds the potential to contribute to personalized health management and enhanced well-being.
However, this study has several limitations. The participant count of 108 was relatively small, preventing full utilization of advanced computational techniques such as deep learning. The cohort was recruited from the Stanford University community and may not represent broader population diversity. Additionally, the average observation period of approximately 1.7 years was too short to track aging patterns over the long term, and molecular data were based solely on blood samples, making it difficult to fully explain direct relationships with specific tissues (e.g., skin, muscle).
Future research should aim to confirm how these nonlinear molecular patterns correspond to functional capacity, disease onset, and changes in mortality risk through long-term longitudinal studies spanning decades with larger, more diverse cohorts, integrating multifaceted data including molecular markers, medical records, functional assessments, and mortality data. Such efforts are essential for deepening our understanding of the complexity of aging and developing innovative strategies for healthy aging.